The Special stains:
Acid-fast stain:
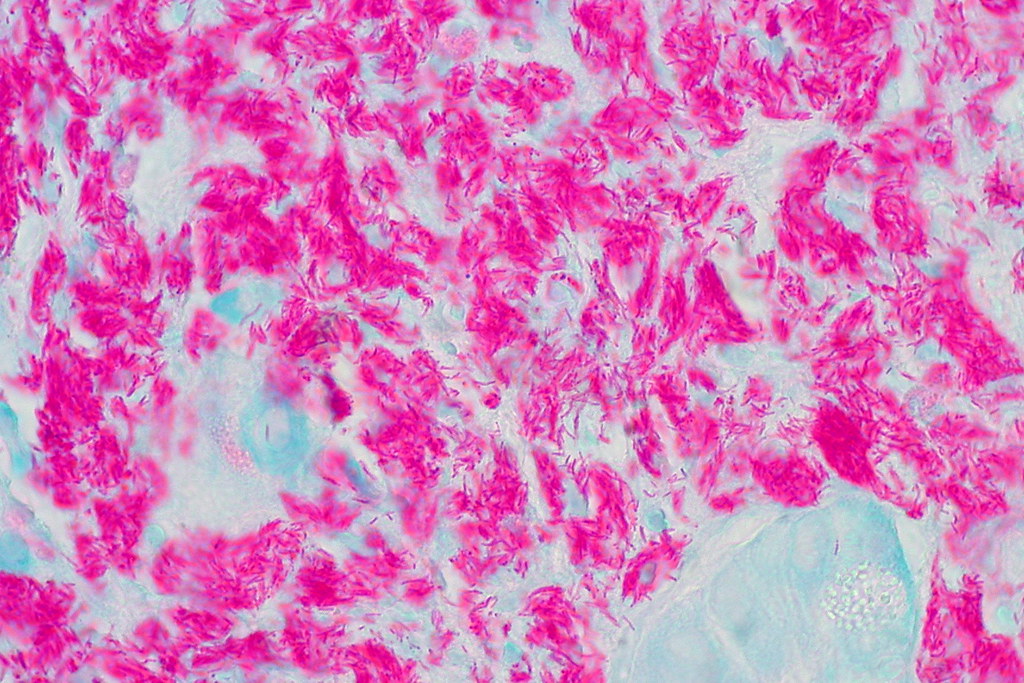
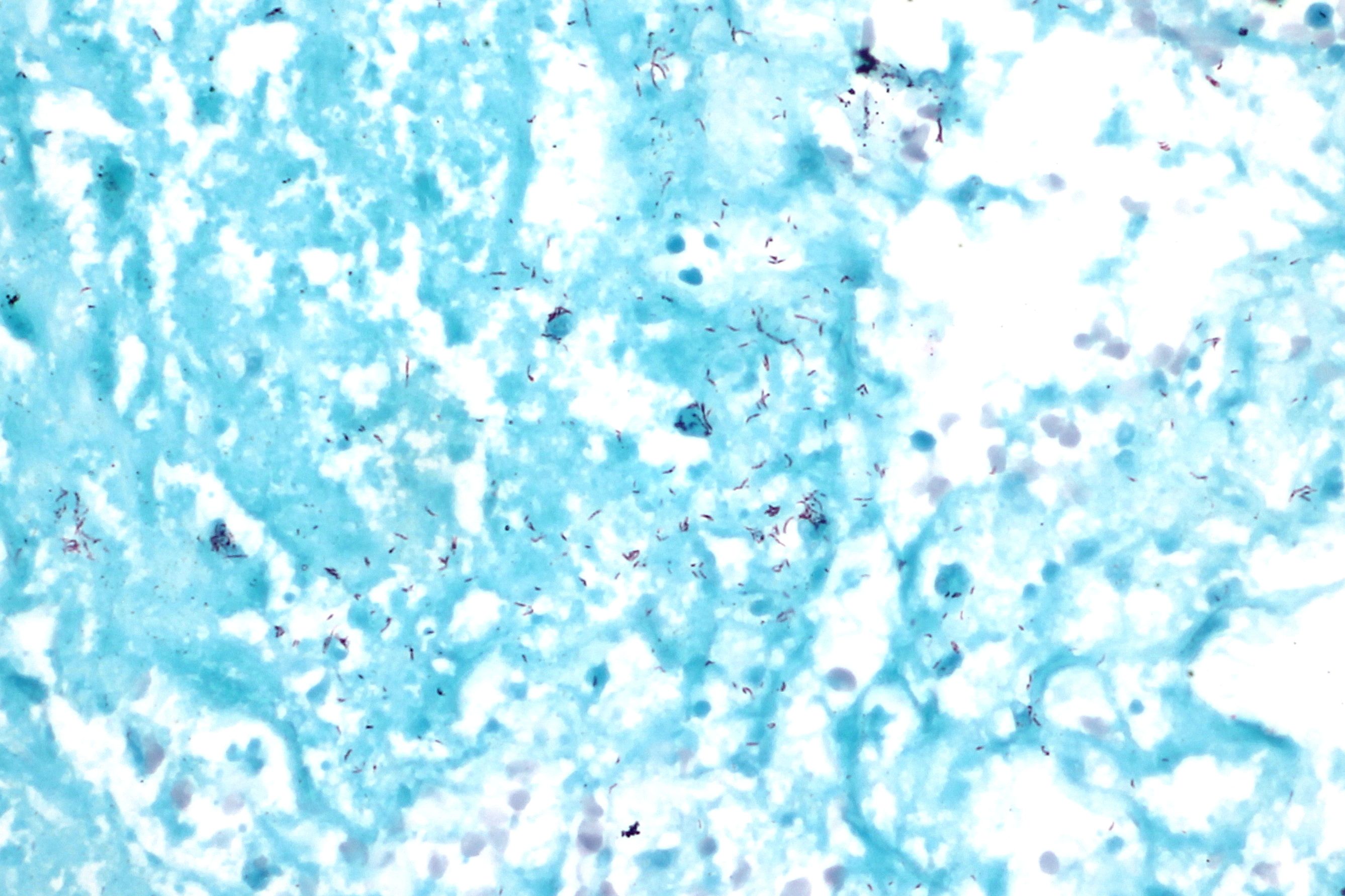
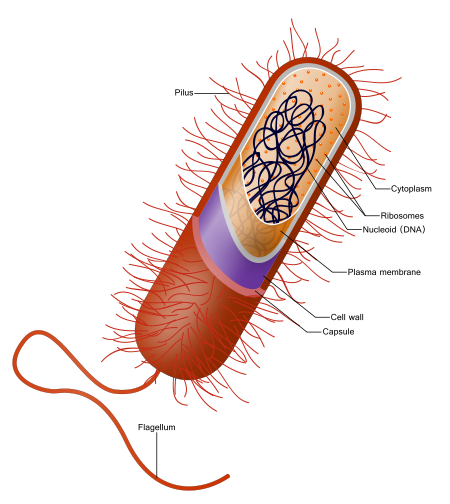
The acid-fast stain is a special type of stain, a differential stain, that is used to identify Mycobacterium (AFB), Actinomycetes and Nocardia species. The cell walls of these types of bacteria are waxy, and will retain the carbolfuchsin primary stain when decolorized with acid-alcohol. Nocardia species are partially acid-fast. There are also a couple of coccidian parasites whose oocysts will also stain with acid-fast stain: Cryptosporidium spp and Cyclospora cayetanensis.
The acid-fast stain aids in the rapid and preliminary diagnosis of the acid-fast bacilli Mycobacterium tuberculosis, or TB, the causative agent of tuberculosis. It has >90% accuracy from sputum samples. It can also be performed to monitor antibiotic therapy for patients with known AFB history or active infection. There are multiple Mycobacterium spp that cause respiratory symptoms similar to TB, as well as multiple other types of infections. See the Mycobacterium tab for information dedicated to this genera.
How does the stain work?
The acid-fast stain works due to the presence of mycolic acids in the cell walls of microbes that are acid-fast. Mycolic acid is waxy and enables the acid-fast microbes to hold/retain the primary phenolic compound carbolfuchsin stain since it is lipid soluble so it penetrates through the waxy cell well, and resist decolorization by the strong acid-alcohol decolorizer. The counterstain is methylene blue or brilliant green. There are two common staining procedures: the Kinyoun (K) or cold method, and the Ziehl-Neelsen (ZN) or hot method. The laboratory where I work utilizes the Kinyoun (K) or cold method. Here is how the methods work:
Kinyoun (K) (Cold Method):
Note: This method is slightly less sensitive than the ZN stain, however, it still produces comparable results.
Ziehl-Neelsen (ZN) (Hot Method):
This method is similar except that involves heat as part of the staining process, whereas the Kinyoun stain is performed completely at room temperature. The two methods produce comparable results, though.
The acid-fast stain aids in the rapid and preliminary diagnosis of the acid-fast bacilli Mycobacterium tuberculosis, or TB, the causative agent of tuberculosis. It has >90% accuracy from sputum samples. It can also be performed to monitor antibiotic therapy for patients with known AFB history or active infection. There are multiple Mycobacterium spp that cause respiratory symptoms similar to TB, as well as multiple other types of infections. See the Mycobacterium tab for information dedicated to this genera.
How does the stain work?
The acid-fast stain works due to the presence of mycolic acids in the cell walls of microbes that are acid-fast. Mycolic acid is waxy and enables the acid-fast microbes to hold/retain the primary phenolic compound carbolfuchsin stain since it is lipid soluble so it penetrates through the waxy cell well, and resist decolorization by the strong acid-alcohol decolorizer. The counterstain is methylene blue or brilliant green. There are two common staining procedures: the Kinyoun (K) or cold method, and the Ziehl-Neelsen (ZN) or hot method. The laboratory where I work utilizes the Kinyoun (K) or cold method. Here is how the methods work:
Kinyoun (K) (Cold Method):
Note: This method is slightly less sensitive than the ZN stain, however, it still produces comparable results.
- Prepare a smear on a slide and allow it to dry completely. For suspected AFB, direct smears are observed first and results are recorded. Samples must be digested and decontaminated and concentrated for the concentrated smear. See the steps on the Mycobacterium tab.
- Flood the slide with lipid-soluble and concentrated primary stain (carbolfuchsin) for 3-4 minutes. Rinse.
- Flood the slide with acid-alcohol decolorizer for 2-3 minutes. Rinse.
- Flood the slide with counterstain (methylene blue or brilliant green) for 4-5 minutes. Rinse
- Completely air dry.
- Observe the entire slide under 40x objective first, then with immersion oil under the 100x objective lens.
- Acid-fast microbes will appear dark/bright pink-red against a blue or blue-green background. Negative cells are blue.
- Always perform a QC slide with your test slide(s) and record your results.
Ziehl-Neelsen (ZN) (Hot Method):
This method is similar except that involves heat as part of the staining process, whereas the Kinyoun stain is performed completely at room temperature. The two methods produce comparable results, though.
- Prepare a smear on a slide and allow it to dry completely. For suspected AFB, direct smears are observed first and results are recorded. Samples must be digested and decontaminated and concentrated for the concentrated smear. See the steps on the Mycobacterium tab.
- Flood the slide with primary stain (carbolfuchsin) for 3-4 minutes and set it on a heat warmer to create steam in order to enhance the stain's ability to enter the cell. Rinse.
- Flood the slide with acid-alcohol decolorizer for 2-3 minutes. Rinse.
- Flood the slide with counterstain (methylene blue or brilliant green) for 4-5 minutes. Rinse
- Completely air dry.
- Observe the entire slide under 40x objective first, then with immersion oil under the 100x objective lens.
- Acid-fast microbes will appear dark/bright pink-red against a blue or blue-green background. Negative cells are blue.
- Always perform a QC slide with your test slide(s) and record your results.
Fluorescent Stain:
Fluorescent staining is typically used for direct smears prior to staining digested, decontaminated samples for concentrated smears. Many laboratories utilize this method in addition to or prior to acid-fast staining.
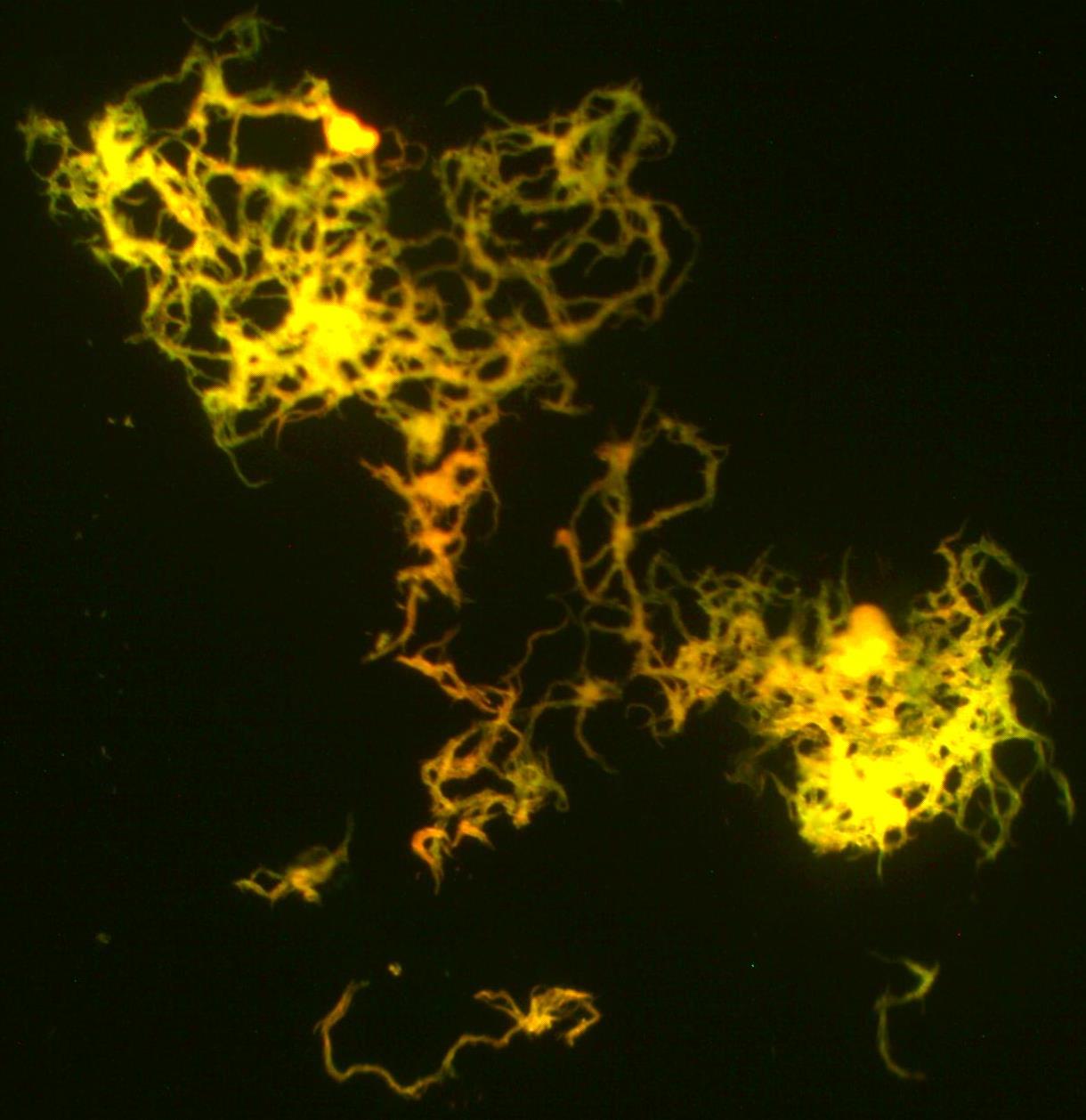
Fluorescent dyes containing fluorescent tags called fluorochromes, such as rhodamine or auramine, which have high sensitivity, bind well with mycolic acid in the cell wall. The cells are then decolorized with an acid-alcohol solution. The cells are counterstained with potassium permanganate. Using a microscope with LED and a blue filter, the acid-fast cells will fluoresce yellow-orange against a dark background.
Fluorescent dyes containing fluorescent tags called fluorochromes, such as rhodamine or auramine, which have high sensitivity, bind well with mycolic acid in the cell wall. The cells are then decolorized with an acid-alcohol solution. The cells are counterstained with potassium permanganate. Using a microscope with LED and a blue filter, the acid-fast cells will fluoresce yellow-orange against a dark background.
- Prepare your smear and let it completely air dry.
- Flood the slide with primary stain (rhodamine or auramine) for 3-4 minutes. Rinse.
- Flood the slide with acid-alcohol decolorizer for 2-3 minutes. Rinse.
- Flood the slide with counterstain potassium permanganate for 4-5 minutes. Rinse.
- Let dry and cover to protect from the light.
- When fully dry, observe the slide under the fluorescent microscope using LED and a blue filter, with the room lights turned down or off.
- Acid-fast bacilli will fluoresce bright yellow-orange against a dark background.
- Always perform a QC slide with your test slide(s) and record your results.
capsule stain:
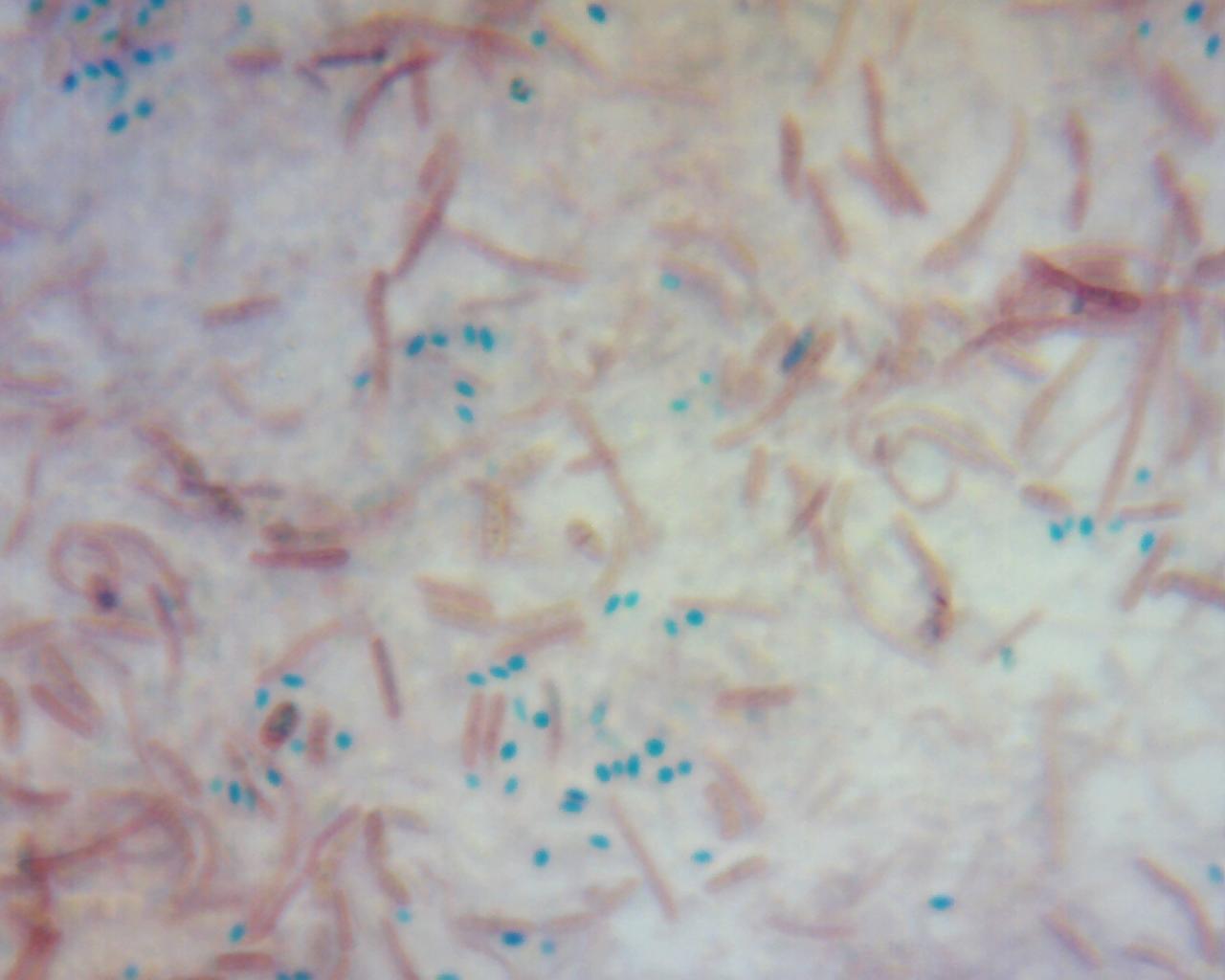
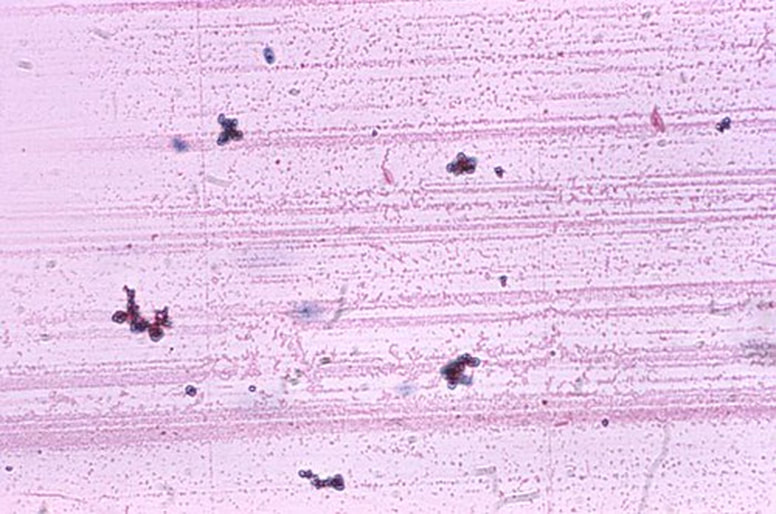
The capsule stain is also a differential stain. It is useful for the detection of cells that produce an extracellular capsule outside the cell wall. This is also sometimes referred to as a "slime capsule". This is a virulence factor in some microbes. It also helps them to evade the immune system and makes antibiotic treatment more difficult, because the capsule is difficult to penetrate with antibacterials.
Capsules are made up of mucoid polysaccharides or polypeptides, which is what effective antibacterials target. These resist most stains so the capsule stain technique stains around the cells, leaving a clearing or "halo" where the capsule is located. The capsule stain is an acidic stain and uses Congo red or nigrosin to stain the background, with a basic stain such as Maneval's that colorizes the cell. The capsule remains unstained. It appears as white, sandwiched between the cells and the background color. The image below is a combination of Congo red and Maneval's stains.
The sample is first spread and mixed in a film of acidic stain and serum and allowed to air dry completely. Do not heat fix the slide, because it will shrink and distort the cells.
Capsules are made up of mucoid polysaccharides or polypeptides, which is what effective antibacterials target. These resist most stains so the capsule stain technique stains around the cells, leaving a clearing or "halo" where the capsule is located. The capsule stain is an acidic stain and uses Congo red or nigrosin to stain the background, with a basic stain such as Maneval's that colorizes the cell. The capsule remains unstained. It appears as white, sandwiched between the cells and the background color. The image below is a combination of Congo red and Maneval's stains.
The sample is first spread and mixed in a film of acidic stain and serum and allowed to air dry completely. Do not heat fix the slide, because it will shrink and distort the cells.
endospore stain:
The endospore stain is also a differential stain. It is useful for detecting the presence of endospores and their location within the cell or if they are free-living. Only a few genera of pathogenic bacteria are spore-forming, and include the Bacillus species and the Clostridium species. Both of these genera of bacteria are mostly soil, freshwater and marine inhabitants, however, some strains live in the human gut and can cause opportunistic infections if the conditions are just right, and others are pathogens:
Clostridium difficile (pseudomembranous colitis)
Clostridium perfringens (gas gangrene and tissue necrosis)
Clostridium botulinum (botulism food poisoning)
Clostridium tetani (tetanus)
Bacillus cereus (food poisoning)
Bacillus anthracis (anthrax)
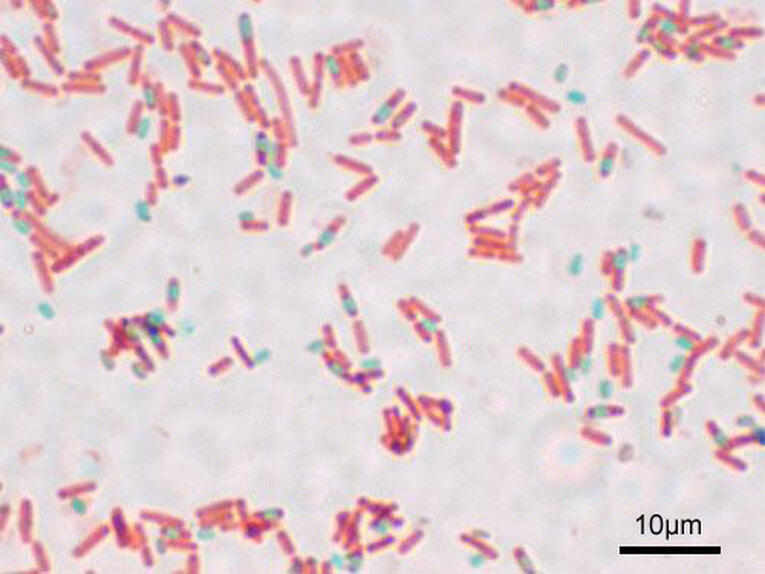
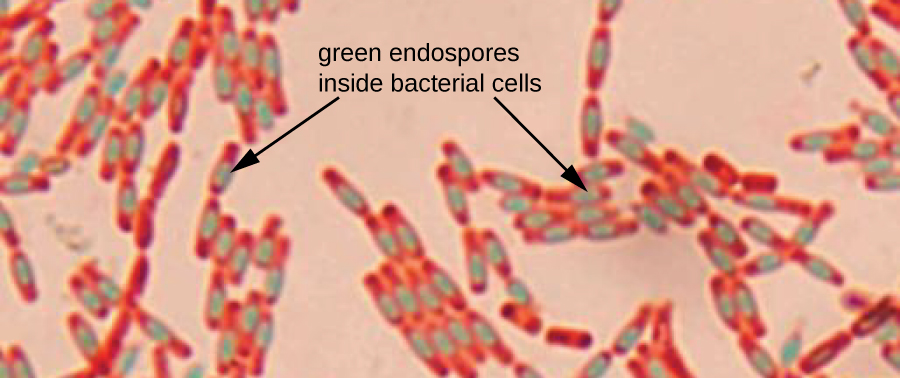
Endospores are are dormant forms of bacterial cells that enable them to survive harsh and extreme conditions, such as extremes of heat, cold, pH, drying, chemicals and others due to their tough outer coating, which is covered with keratin protein, the same protein found in hair and nails. Keratin resists staining. It takes extreme techniques to stain the spores. The endospore staining method is called the Schaeffer-Fulton method. A primary stain of malachite green is applied first, which is forced into the spore via steam or left on the slide for 15+ minutes to penetrate the spore. It is water-soluble and has a low affinity for the vegetative cell that is housing the spore so the mother cell will be decolorized with water and then counterstained with safranin.
Spores can be found in various locations within the cell. They may be found in the middle (central), at the end (terminal) or between the middle and the end of the cell (subterminal) and are round-to-oval. Some are small and others give the cell a "swollen" appearance. In this method, the spores will appear green and the vegetative mother cells will be pink-red.
Clostridium difficile (pseudomembranous colitis)
Clostridium perfringens (gas gangrene and tissue necrosis)
Clostridium botulinum (botulism food poisoning)
Clostridium tetani (tetanus)
Bacillus cereus (food poisoning)
Bacillus anthracis (anthrax)
Endospores are are dormant forms of bacterial cells that enable them to survive harsh and extreme conditions, such as extremes of heat, cold, pH, drying, chemicals and others due to their tough outer coating, which is covered with keratin protein, the same protein found in hair and nails. Keratin resists staining. It takes extreme techniques to stain the spores. The endospore staining method is called the Schaeffer-Fulton method. A primary stain of malachite green is applied first, which is forced into the spore via steam or left on the slide for 15+ minutes to penetrate the spore. It is water-soluble and has a low affinity for the vegetative cell that is housing the spore so the mother cell will be decolorized with water and then counterstained with safranin.
Spores can be found in various locations within the cell. They may be found in the middle (central), at the end (terminal) or between the middle and the end of the cell (subterminal) and are round-to-oval. Some are small and others give the cell a "swollen" appearance. In this method, the spores will appear green and the vegetative mother cells will be pink-red.
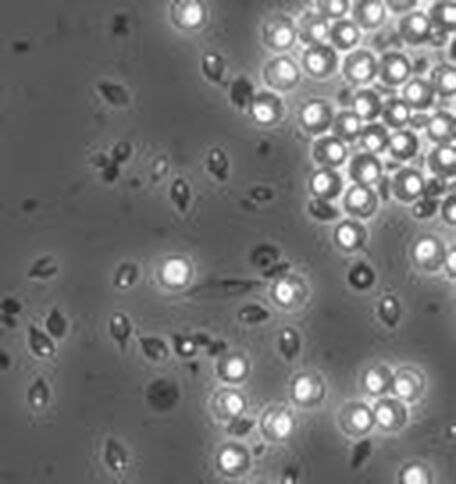
phase contrast microscopy:
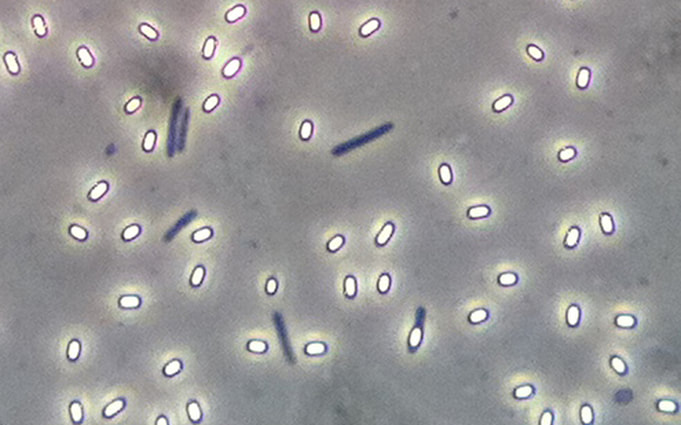
Phase contrast microscopy can also be used to visualize spores and other cells. In these images phase contrast microscopy reveals free spores, the shape of the spores, spore location within cells, some are round, some are elliptical, some are at the end of the vegetative mother cell, and others are in the middle.
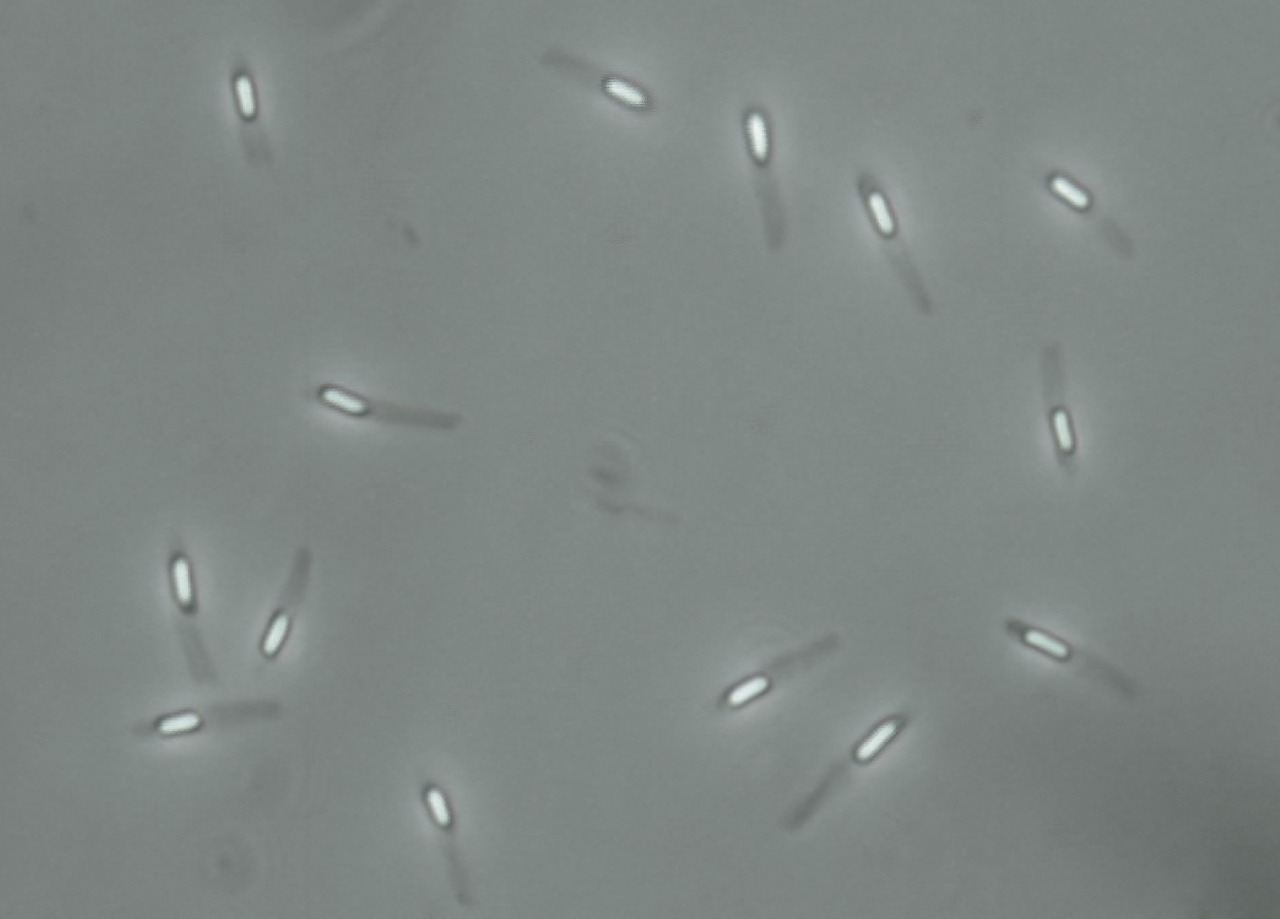
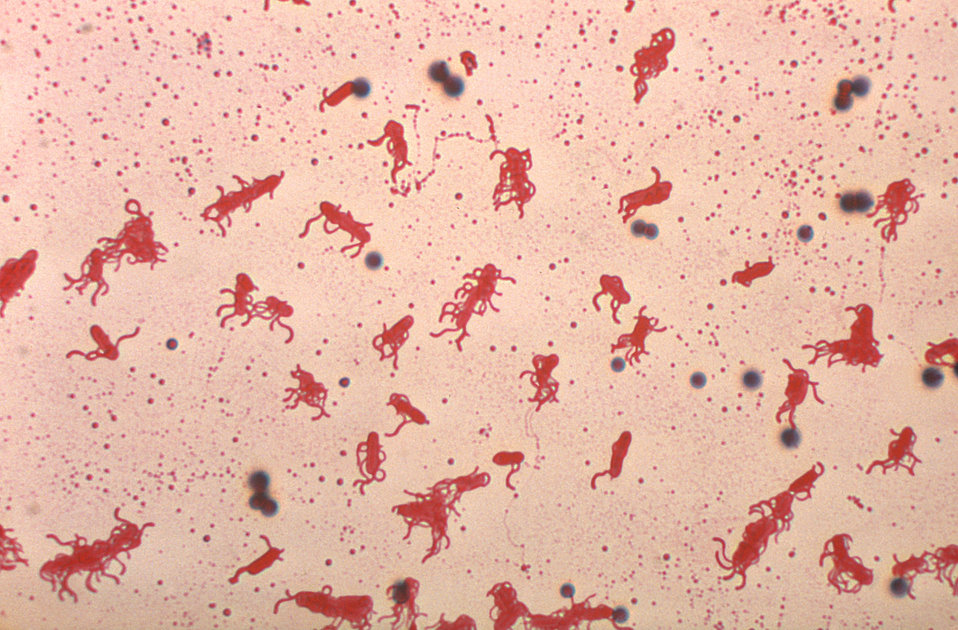
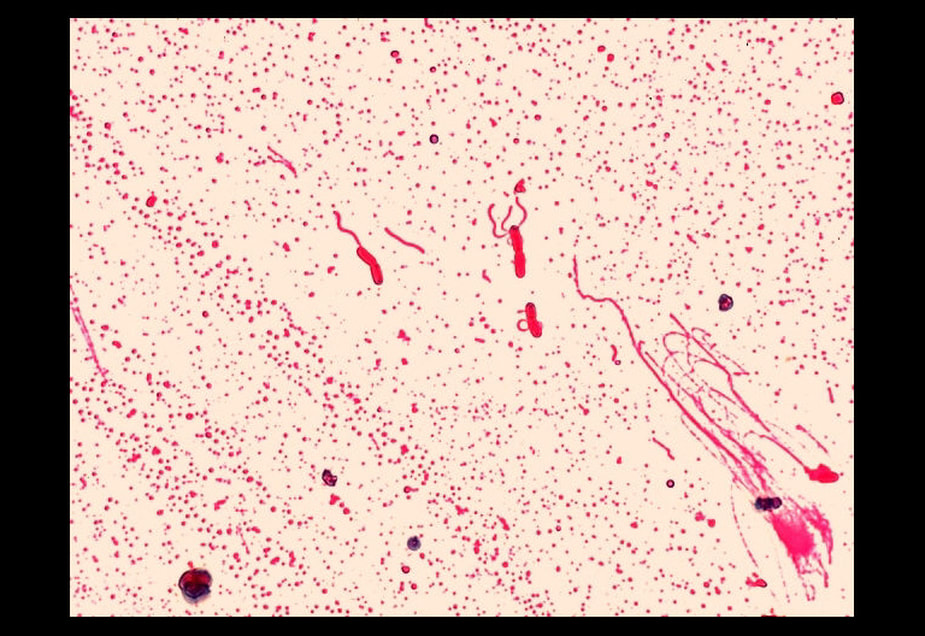
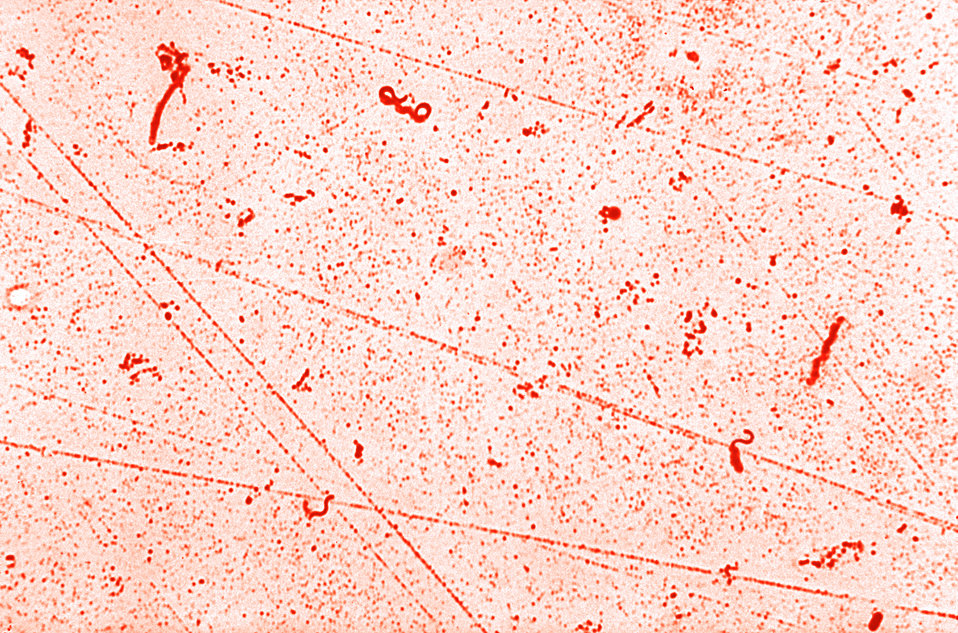
flagella stain:
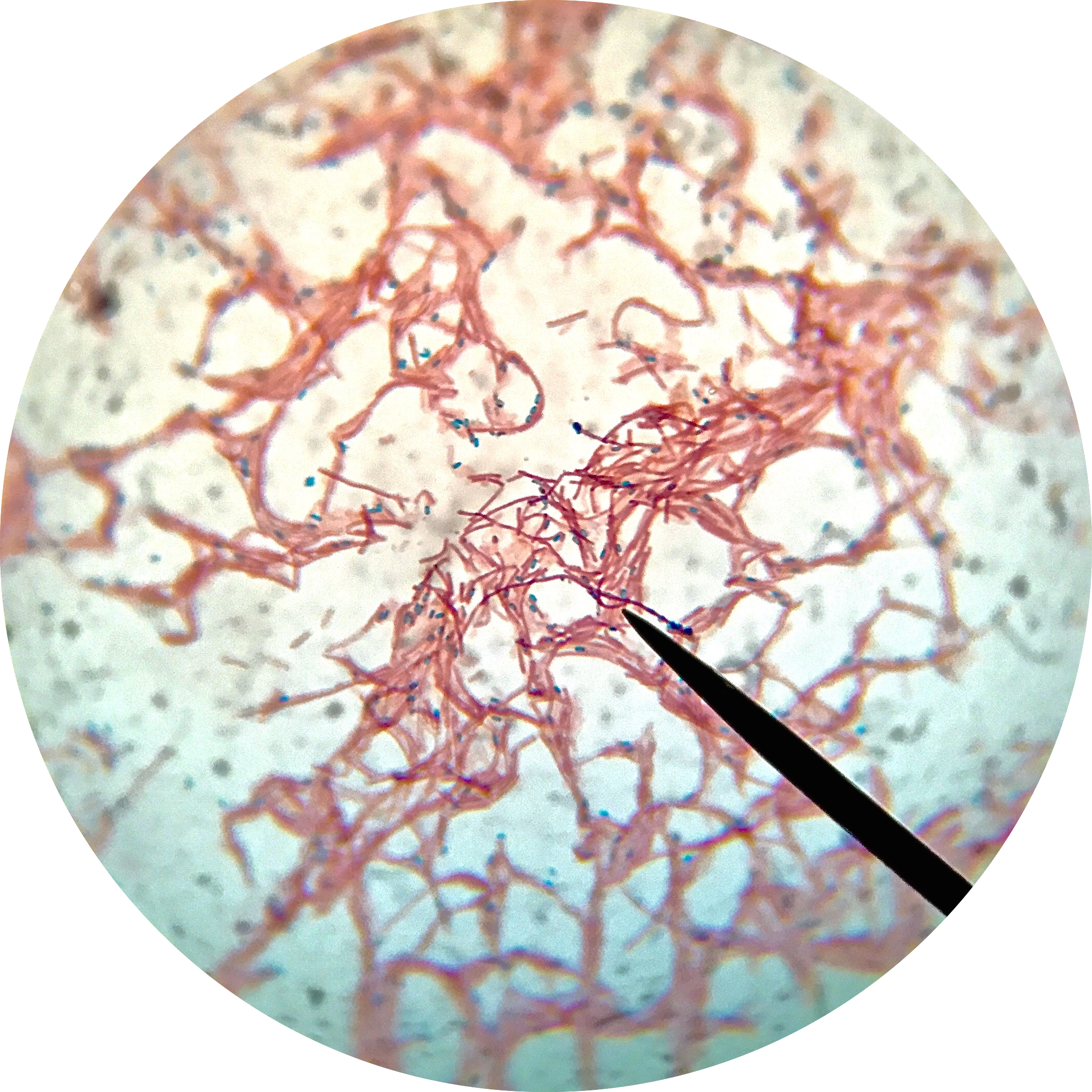
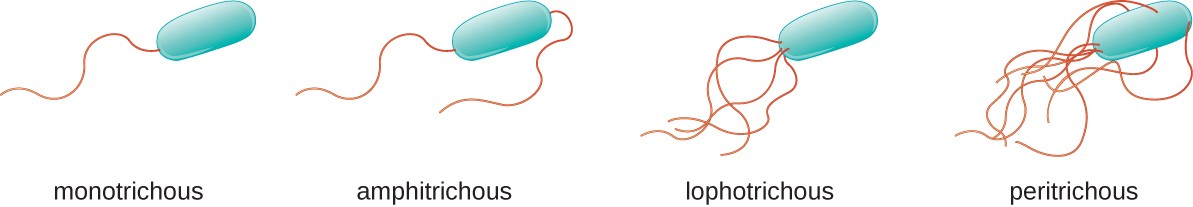
The flagella stain enables direct observation of flagella and their arrangement or morphology. This may aid in the identification of bacterial cells. There are 4 classes of flagella:
The flagella stain is a special stain. It uses a mordant to assist in helping the stain to stick to the flagella so that it will be visible.
Examples:
- Monotrichous (single polar)
- Amphitrichous (bipolar, one at each end)
- Lophotrichous (tuft at one end)
- Peritrichous (all over the surface)
The flagella stain is a special stain. It uses a mordant to assist in helping the stain to stick to the flagella so that it will be visible.
Examples:
- Unipolar, monotrichous: Pseudomonas aeruginosa, Campylobacter species
- Bipolar, amphitrichous: Spirillum spp
- Tuft, lophotrichous: Pseudomonas species (other)
- All over, peritrichous: Proteus vulgaris (swarmer), Escherichia coli (gut)