biochemical tests and assays used in bacteriology:
catalase slide test:
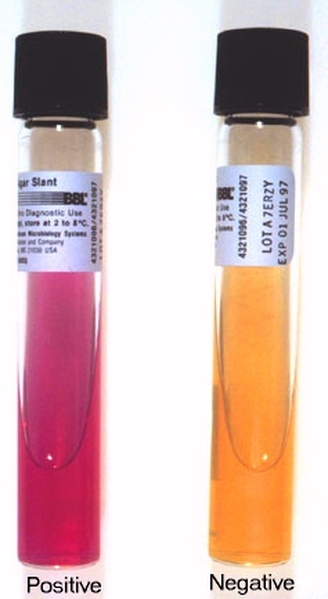
CATALASE TEST: A positive catalase test is shown in the image above.
The catalase test is a simple test used by microbiologists to help identify and differentiate between species and strains of bacteria and to determine the ability of some microbes to break down hydrogen peroxide by producing the enzyme catalase. Hydrogen peroxide (3%) is used to detect the presence of catalase enzyme in the bacterial isolate that is being tested. A small quantity of bacteria is added to a drop of hydrogen peroxide on the slide. Bubbles of oxygen will be observed if the bacteria possess the catalase enzyme, because the enzyme catalyzes the break down of hydrogen peroxide into oxygen and water, and you will observe this reaction occur. If bubbles are observed upon the addition of bacteria to the hydrogen peroxide, the organism is said to be catalase positive (example: Staphylococcus aureus). If no bubbles are observed when bacteria is added to the drop of hydrogen peroxide on the slide, the organism is said to be catalase negative (example: Streptococcus pyogenes). If very mild to weak bubbling is observed, it is said to be weakly catalase positive (example: Enterococcus faecalis).
Principle of the Catalase Test:
Some microbes produce hydrogen peroxide during aerobic respiration (in the presence of oxygen) and, in some cases, a very potent superoxide. A build up of these substances will result in death of the organism unless they can be degraded or broken down enzymatically. Aerobes, facultative anaerobes, and microaerophiles use the aerobic respiration pathway, in which oxygen is the final electron acceptor (ETC, or electron transport chain), during the break down of carbohydrates for the production of energy. Organisms that are able to produce the enzyme catalase quickly break down hydrogen peroxide into water and free oxygen.
How to Perform the Catalase Test:
The catalase test is a simple test used by microbiologists to help identify and differentiate between species and strains of bacteria and to determine the ability of some microbes to break down hydrogen peroxide by producing the enzyme catalase. Hydrogen peroxide (3%) is used to detect the presence of catalase enzyme in the bacterial isolate that is being tested. A small quantity of bacteria is added to a drop of hydrogen peroxide on the slide. Bubbles of oxygen will be observed if the bacteria possess the catalase enzyme, because the enzyme catalyzes the break down of hydrogen peroxide into oxygen and water, and you will observe this reaction occur. If bubbles are observed upon the addition of bacteria to the hydrogen peroxide, the organism is said to be catalase positive (example: Staphylococcus aureus). If no bubbles are observed when bacteria is added to the drop of hydrogen peroxide on the slide, the organism is said to be catalase negative (example: Streptococcus pyogenes). If very mild to weak bubbling is observed, it is said to be weakly catalase positive (example: Enterococcus faecalis).
Principle of the Catalase Test:
Some microbes produce hydrogen peroxide during aerobic respiration (in the presence of oxygen) and, in some cases, a very potent superoxide. A build up of these substances will result in death of the organism unless they can be degraded or broken down enzymatically. Aerobes, facultative anaerobes, and microaerophiles use the aerobic respiration pathway, in which oxygen is the final electron acceptor (ETC, or electron transport chain), during the break down of carbohydrates for the production of energy. Organisms that are able to produce the enzyme catalase quickly break down hydrogen peroxide into water and free oxygen.
How to Perform the Catalase Test:
- Place one drop of hydrogen peroxide on a clean microscope slide.
- Using an applicator stick or a loop, touch 1-3 of the most isolated colonies, and smear a sample into the hydrogen peroxide drop.
- Observe the mixture for the rapid production of bubbles or froth (within 5-10 seconds). If the mixture produces many bubbles or froth, the organism is catalase positive.
- A negative catalase test result is no bubbles or only a few scattered bubbles (up to about 20 seconds).
- A false positive catalase test result may occur if you use a metal loop or needle with hydrogen peroxide, since it degrades the metal. If using colonies from a blood agar plate and you accidentally scrape part of the blood agar, this may yield a false positive test, as damaged sheep's red blood cells in the agar release catalase, which will react with the hydrogen peroxide. Finally, because some bacteria do possess enzymes other than catalase that can break down hydrogen peroxide, a few small bubbles that form after 20-30 seconds may not be considered a positive test.
- Examples of catalase positive organisms: Staphylococci, Micrococci, Listeria, Corynebacterium, Nocardia, Burkholderia cepacia, Citrobacter, Escherichia coli, Enterobacter, Klebsiella, Shigella, Yersinia, Proteus, Salmonella, Serratia, Pseudomonas, Rhodococcus equi, Mycobacterium tuberculosis, Legionella pneumophila, Campylobacter jejuni, Bacillus spp
- Examples of catalase negative organisms: Streptococci, Enterococcus (with the exception of Enterococcus faecalis, which is weakly catalase positive).
agglutination tests:
There are a variety of agglutination tests used in the microbiology laboratory to aid in the identification of bacteria. Agglutination reactions are used to detect the presence of an antigen or the presence of an antibody or a complex between the two. Direct agglutination reactions can help in the diagnosis of some diseases. They can also help determine if an individual was exposed to a pathogen. They are also used in Blood Banking to determine the blood type. Indirect agglutination reactions, on the other hand, are also used in some disease diagnoses and are the principle behind pregnancy tests.
How do these tests work? Particulate antigens are able to combine with antibodies to form visible clumps called agglutinins or agglutinates. Agglutination is the term for this clumping. This is proof that an antigen-antibody complex has formed and this means you have a positive result.
Direct Agglutination: the combination of antibodies and naturally particulate antigens
Indirect Agglutination: artificial or synthetically made systems (such as latex particles) are used to detect agglutination by coating particles like latex microspheres with antibody OR antigen, where it will result in visible clumping
Slide agglutination tests are examples of direct agglutination tests by mixing samples of antigen and antiserum on a slide to see if there is clumping (a reaction). Visible clumps mean a positive result.
How do these tests work? Particulate antigens are able to combine with antibodies to form visible clumps called agglutinins or agglutinates. Agglutination is the term for this clumping. This is proof that an antigen-antibody complex has formed and this means you have a positive result.
Direct Agglutination: the combination of antibodies and naturally particulate antigens
Indirect Agglutination: artificial or synthetically made systems (such as latex particles) are used to detect agglutination by coating particles like latex microspheres with antibody OR antigen, where it will result in visible clumping
Slide agglutination tests are examples of direct agglutination tests by mixing samples of antigen and antiserum on a slide to see if there is clumping (a reaction). Visible clumps mean a positive result.
coagulase slide test (latex agglutination) and coagulase tube test:
THE LATEX AGGLUTINATION TEST:
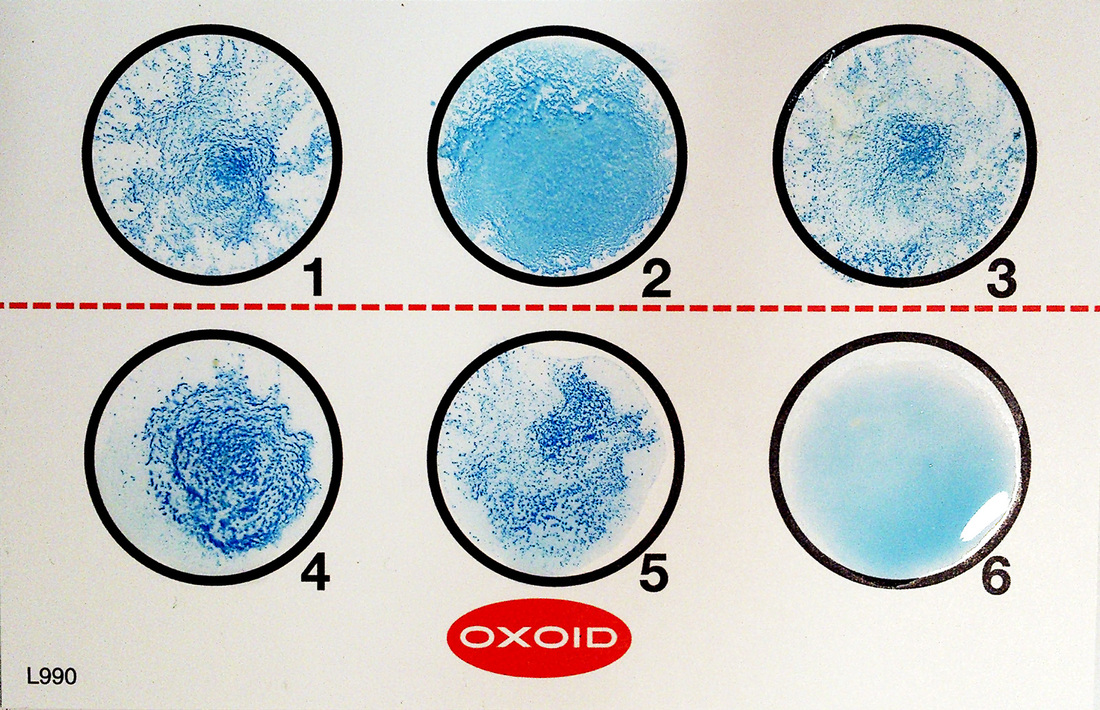
The latex slide agglutination test is a rapid test used to aid in the differentiation of Staphylococcus aureus from other staphylococcus species. This particular kit uses blue latex particles that are coated with porcine fibrinogen and rabbit IgG (immunoglobulin), including specific polyclonal antibodies raised against capsular polysaccharides of Staphylococcus aureus. A positive result is seen in the image above as specific clumping after rocking the slide for 1 minute for samples 1-5, and the negative sample No. 6.
THE COAGULASE TEST (See 2 Images Below):
Staphylococcus aureus produces two forms of the enzyme coagulase: bound coagulase and free coagulase.
Bound coagulase, or "clumping factor", can be detected by performing a slide coagulase test.
Free coagulase can be detected by performing a tube coagulase test, as seen in the image below and discussed below.
The coagulase tests are based on the principle that the enzyme coagulase enables the conversion of fibrinogen to fibrin, resulting in the formation of a clot. Coagulase enzyme reacts with prothrombin in the blood. It forms a complex known as staphylothrombin. This complex enables the enzyme protease to convert fibrinogen (plasma protein produced by the liver) into fibrin, which results in the clotting of the blood (coagulation). This enzyme is tightly bound to the surface of Staphylococcus aureus. When S. aureus comes into contact with blood, coagulase enzyme can coat its surface with fibrin. The fibrin clot is able to protect the bacterium from the immune system and from destruction by phagocytosis, making this bacterium even more virulent.
Additionally, about 95% of S. aureus bacteria affecting humans possess Protein A, which binds tightly to the Fc portion of the IgG antibody. Therefore, the slide coagulase test consists of latex particles coated with both human fibrinogen AND IgG. If one or both of these are present, there will be visible, macroscopic clumping, and agglutination will occur and the test will be positive within 20 seconds with no agglutination in the control circle. If neither one is present, there will be no visible clumping. No agglutination will be seen in the test circle and the test will be negative.
Reagents contain sodium azide, which is toxic upon inhalation and very toxic to the skin and a corrosive.
Things to Consider When Using the Staphylococcal Coagglutination Procedure:
-Hyperproteinemia may cause autoagglutination and false-positive test results (example: S. saprophyticus will agglutinate the test reagent and the control and this is a false + due to autoagglutination)
-Always shake the test latex reagents really good to reduce settling of the latex antibodies prior to performing the test
THE COAGULASE SLIDE TEST (Image Below: Upper Right):
The latex slide agglutination test is a rapid test used to aid in the differentiation of Staphylococcus aureus from other staphylococcus species. This particular kit uses blue latex particles that are coated with porcine fibrinogen and rabbit IgG (immunoglobulin), including specific polyclonal antibodies raised against capsular polysaccharides of Staphylococcus aureus. A positive result is seen in the image above as specific clumping after rocking the slide for 1 minute for samples 1-5, and the negative sample No. 6.
THE COAGULASE TEST (See 2 Images Below):
Staphylococcus aureus produces two forms of the enzyme coagulase: bound coagulase and free coagulase.
Bound coagulase, or "clumping factor", can be detected by performing a slide coagulase test.
Free coagulase can be detected by performing a tube coagulase test, as seen in the image below and discussed below.
The coagulase tests are based on the principle that the enzyme coagulase enables the conversion of fibrinogen to fibrin, resulting in the formation of a clot. Coagulase enzyme reacts with prothrombin in the blood. It forms a complex known as staphylothrombin. This complex enables the enzyme protease to convert fibrinogen (plasma protein produced by the liver) into fibrin, which results in the clotting of the blood (coagulation). This enzyme is tightly bound to the surface of Staphylococcus aureus. When S. aureus comes into contact with blood, coagulase enzyme can coat its surface with fibrin. The fibrin clot is able to protect the bacterium from the immune system and from destruction by phagocytosis, making this bacterium even more virulent.
Additionally, about 95% of S. aureus bacteria affecting humans possess Protein A, which binds tightly to the Fc portion of the IgG antibody. Therefore, the slide coagulase test consists of latex particles coated with both human fibrinogen AND IgG. If one or both of these are present, there will be visible, macroscopic clumping, and agglutination will occur and the test will be positive within 20 seconds with no agglutination in the control circle. If neither one is present, there will be no visible clumping. No agglutination will be seen in the test circle and the test will be negative.
Reagents contain sodium azide, which is toxic upon inhalation and very toxic to the skin and a corrosive.
Things to Consider When Using the Staphylococcal Coagglutination Procedure:
-Hyperproteinemia may cause autoagglutination and false-positive test results (example: S. saprophyticus will agglutinate the test reagent and the control and this is a false + due to autoagglutination)
-Always shake the test latex reagents really good to reduce settling of the latex antibodies prior to performing the test
THE COAGULASE SLIDE TEST (Image Below: Upper Right):
- A slide coagulase test is run with a negative control to rule out autoagglutination.
- Two drops of saline are put onto the slide next to each other, one labeled with sample number (the test) and one labeled as the control (C).
- Each of the two saline drops are emulsified with the test organism using a wire loop, straight wire, or wooden stick.
- One drop of plasma rabbit plasma is added to the inoculated saline drop corresponding to the sample test and mixed well.
- The slide is rocked gently for about 10-20 seconds.
- If clumping is observed in the plasma within the 10-20 seconds, with no clumping in the saline drop, the test is said to be coagulase positive.
- If no clumping is observed within the plasma or the saline drop within 20 seconds, the test is said to be coagulase negative.
- If the slide coagulase test is negative, a coagulase tube test should follow to confirm it.
- If clumping occurs in both the plasma drop and the saline drop, this is an indication of autoagglutination, and a coagulase tube test should be performed.
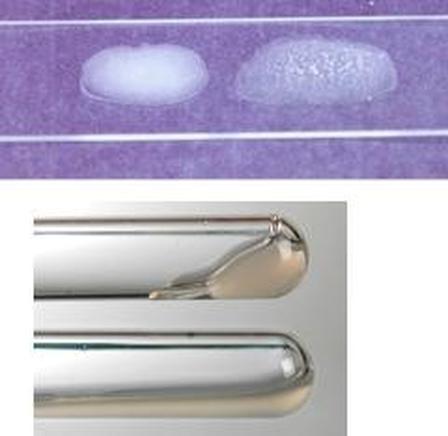
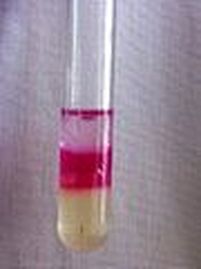
Positive Tube Coagulase Test is seen in the image above (Left and Lower Right)
THE COAGULASE TUBE TEST (See Image Above on the Left and Lower Right):
THE COAGULASE TUBE TEST (See Image Above on the Left and Lower Right):
- The coagulase tube test uses rabbit plasma that has been inoculated with Staphylococcus aureus in a test tube.
- The tube is incubated at 37 degrees Celsius for 1.5 hours.
- If the test is negative (no clumping observed at 1.5 hours), then incubation shall continue for up to 18 hours.
- A fibrin clot will be observed in the bottom of the test tube by the coagulase reaction of a positive test. The clot may be so thick that the rest of the liquid may totally solidify.
- If the test is negative, the plasma will remain as liquid.
- The test must be allowed to cool at room temperature for 30 minutes or in the freezer for 10 minutes prior to reading, otherwise you may get a false negative result. Serum can melt, so it is important to make sure that the serum remains liquid after cooling for a true negative result.
- Coagulase-positive staphylococci: Staphylococcus aureus subsp. anaerobius, S. a. aureus, S. a. delphini, S. hyicus, S. intermedius, S. lutrae, and Staphylococcus schleiferi subsp. coagulans.
- Coagulase-negative staphylococci of clinical significance: S. saprophyticus, S.cohnii subsp. cohnii, S. cohnii subsp. urealyticum, S. captitus subsp. captitus, S. warneri, S.hominis, S.epidermidis, S. caprae, and S.lugdunensis

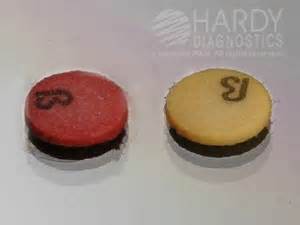
The image on the left shows another method for the coagulase slide test. Instead of using a glass slide, a card like this one may be used with a black background, which enables the observer to see the clumping more clearly. Circle 1 shows a positive coagulase slide test (clumping), and circle 2 shows a negative reaction (no clumping).
streptococcus typing (groups a, b, c, f, g):
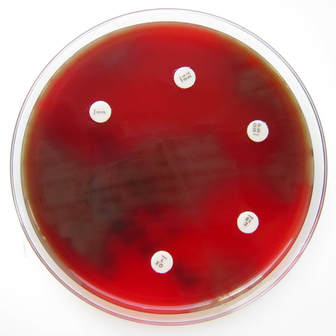
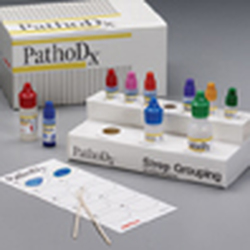
THE PATHODX STREPTOCOCCUS TYPING KIT (See Image Below):
The Remel PathoDx Kit is a latex slide agglutination test used to rapidly and accurately identify beta-hemolytic streptococci groups A, B, C, F and G. The latex agglutination test allows for flexible procedure options that allow for 100% correlation to the Lancefield precipitin method. Care should be taken to wear proper PPE when performing this test, because the reagents are caustic and eye irritants.
This test is based on the antigenic characteristics of the C carbohydrate (sugar) on the cell wall, or Lancefield antigens, of which there are many, but we focus on A, B, C, F and G here.
Lancefield Antigens:
1) Group A: Streptococcus pyogenes ("fever-producing"), causative agent of streptococcal pharyngitis, or "Strep Throat"
2) Group B: Streptococcus agalactiae, which can colonize women of child-bearing age, and can cause neonatal meningitis in infants after birth
3) Group C: S. equi, S. dysgalactiae
4) Group D: (Not tested for with this kit, but includes the Enterococci, S. bovis, S. equinus)
5) Group F: S. anginosus, S. milleri
6) Group G: S. canis
Reagents in the Kit:
1) Sodium nitrate: Reagent 1
2) Glacial Acetic Acid: Reagent 2
3) Sodium Carbonate: Reagent 3
4) Grouping Latex A
5) Positive and Negative Controls
6) Grouping Latex B, C, F, G
The first three reagents are kept at room temperature. The Grouping Latex and Positive and Negative Controls are kept in the refrigerator at up to 8 degrees Celsius.
How to Perform the Test:
The Remel PathoDx Kit is a latex slide agglutination test used to rapidly and accurately identify beta-hemolytic streptococci groups A, B, C, F and G. The latex agglutination test allows for flexible procedure options that allow for 100% correlation to the Lancefield precipitin method. Care should be taken to wear proper PPE when performing this test, because the reagents are caustic and eye irritants.
This test is based on the antigenic characteristics of the C carbohydrate (sugar) on the cell wall, or Lancefield antigens, of which there are many, but we focus on A, B, C, F and G here.
Lancefield Antigens:
1) Group A: Streptococcus pyogenes ("fever-producing"), causative agent of streptococcal pharyngitis, or "Strep Throat"
2) Group B: Streptococcus agalactiae, which can colonize women of child-bearing age, and can cause neonatal meningitis in infants after birth
3) Group C: S. equi, S. dysgalactiae
4) Group D: (Not tested for with this kit, but includes the Enterococci, S. bovis, S. equinus)
5) Group F: S. anginosus, S. milleri
6) Group G: S. canis
Reagents in the Kit:
1) Sodium nitrate: Reagent 1
2) Glacial Acetic Acid: Reagent 2
3) Sodium Carbonate: Reagent 3
4) Grouping Latex A
5) Positive and Negative Controls
6) Grouping Latex B, C, F, G
The first three reagents are kept at room temperature. The Grouping Latex and Positive and Negative Controls are kept in the refrigerator at up to 8 degrees Celsius.
How to Perform the Test:
- Take the refrigerated reagents out of the refrigerator and allow to come to room temperature.
- Label one test tube as "positive control", one as "negative control", and each of your tests with the requisition or sample ID number.
- Put 2 drops of Reagent 1 in the test tube.
- Put 2 drops of Reagent 2 in the test tube.
- Touch the tops of approximately 5-6 of the purest, most isolated bacterial colonies with the wooden applicator stick and emulsify with the reagents in the test tube.
- Add 4 drops of Reagent 3 to the test tube, which will bubble. Mix well.
- Label the slide card circles as "positive control", "negative control", and with each of your test sample ID numbers.
- Put one drop of the positive control in the circle labeled "positive".
- Put one drop of the negative control in the circle labeled "negative".
- Put one drop of the test specimen into the circle on the test card.
- Shake the Grouping Latex A to make sure that you have a thorough mixing of the reagent and reduce the chance for false clumping.
- Opposite of the drop, place one drop of the Grouping Latex A (it will be blue).
- Mix and spread the 2 drops together over the entire circle, using a separate wooden applicator stick to mix each circle.
- Rock the slide gently for 2 minutes and observe for agglutination.
- If specific agglutination is observed, the test is considered to be positive for Group A Streptococccus (GAS).
- If no agglutination is observed, the test is considered to be negative for Group A Streptococcus.
- If the beta-hemolytic colony growth on the plate was heavy (into quadrant 4), and the Grouping Latex A test was negative for agglutination, a Grouping Test with B, C, F and G should be performed.
enzyme-linked immunosorbent assay (elisa) tests:
Many of the rapid immunoassay tests in the microbiology lab use this technique, such as cards used to detect things like stool samples for Shigella, etc...
ELISA is used to detect the presence as well as the amount of antigen OR antibody present in a specimen. Indirect ELISA tests are rapid tests used to screen patients for things like HIV antibodies, rubella virus antibodies, Giardia, Cryptosporidium, Infectious Mononucleosis, Strep Throat, Influenza, RSV, and others. Direct ELISA tests are used to detect hormones like HCG in some types of pregnancy tests or LH in ovulation tests, drugs, bacterial antigens, or viral antigens.
Direct ELISA Method:
ELISA is used to detect the presence as well as the amount of antigen OR antibody present in a specimen. Indirect ELISA tests are rapid tests used to screen patients for things like HIV antibodies, rubella virus antibodies, Giardia, Cryptosporidium, Infectious Mononucleosis, Strep Throat, Influenza, RSV, and others. Direct ELISA tests are used to detect hormones like HCG in some types of pregnancy tests or LH in ovulation tests, drugs, bacterial antigens, or viral antigens.
Direct ELISA Method:
- Detects the presence of antigen in the sample
- Antibody (antiserum) specific for that antigen is coated (adsorbed) onto the wall of a microtiter well
- The sample is added
- If the antigen is present, it will bind to the antibody, and they will form a complex
- Another antibody with an enzyme attached to it specific for the same antigen is added, and any unbound enzyme-linked antibody is washed away
- Substrate for the enzyme is added and it is converted to product, indicated by a color change, which means the sample contains the antigen
- No color change is a negative result
- Detects the presence of antibody in the sample
- The antigen specific for the antibody is coated (adsorbed) onto the wall of the microtiter well
- The sample is added
- If antibody is present, it will bind to the antigen
- An antihuman (synthetic) immunoglobulin antibody with an attached enzyme is added
- Unbound enzyme-linked antibody is washed away
- Substrate for the enzyme is added and it is converted to product, indicated by a color change, which means the sample contains the antibody
- No color change is a negative result
fluorescent antibody (fa) technique:
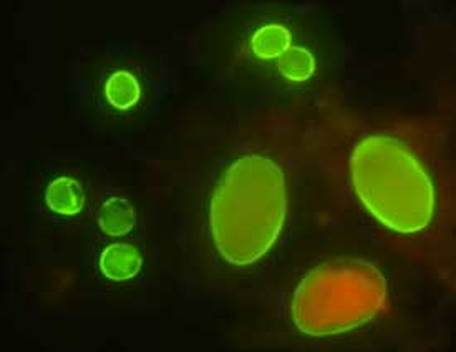
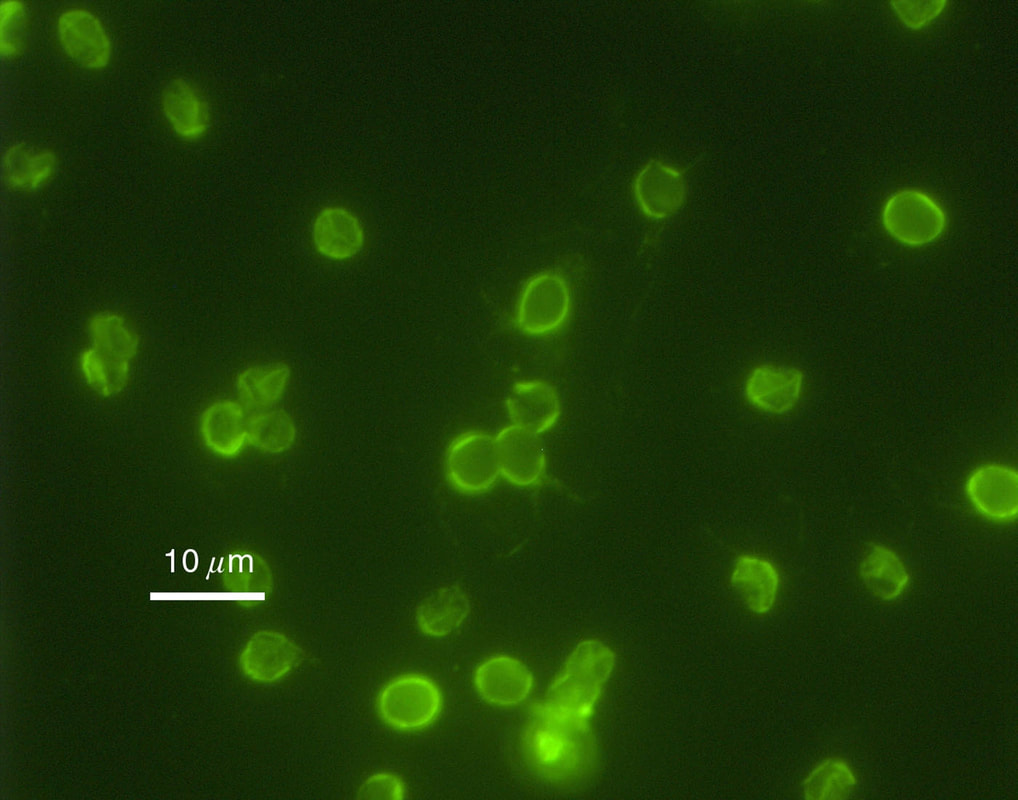
Fluorescence tests are useful in identifying many fungi, parasites, and viruses. This type of testing uses fluorescent tags to indicate the presence of antigen or antibody in a sample. Direct tests (DFA) identify the presence of antigens, whereas Indirect tests (IFA) identify the presence of antibody in the sample.
Fluorescent antibodies are labeled with a tag (fluorescein isothiocyanate dye). It fluoresces when illuminated with UV light. In a DFA, if the sample contains a suspected antigen and it is fixed to a microscope slide, the fluorescent antibody is then added and given time to react with the antigen to form a complex. The slide is rinsed to remove unbound antibody. The slide is then observed with a fluorescent microscope fitted with a UV light source. If the antigen is present, the bound antibodies will complex to emit a bright apple green fluorescent color.
In an IFA, the same process is used, but it is used to detect antibodies rather than antigens in a sample. In this case, the antigen is fixed to the slide and dilutions of patient samples are fixed to several slides and incubated in the dark at room temperature to give them time to react with the antigen. The FITC-labeled antibody is an anti-gamma globulin antibody. This means that if there is any patient antibody bound to antigen on the slide, the fluorescent antibody will bind to it. The slides are rinsed to remove any unbound fluorescent antibodies. The slides are now ready to view under the microscope with a UV light source. If the antibody is present, the labeled antibodies will fluoresce a bright apple green.
Fluorescent antibodies are labeled with a tag (fluorescein isothiocyanate dye). It fluoresces when illuminated with UV light. In a DFA, if the sample contains a suspected antigen and it is fixed to a microscope slide, the fluorescent antibody is then added and given time to react with the antigen to form a complex. The slide is rinsed to remove unbound antibody. The slide is then observed with a fluorescent microscope fitted with a UV light source. If the antigen is present, the bound antibodies will complex to emit a bright apple green fluorescent color.
In an IFA, the same process is used, but it is used to detect antibodies rather than antigens in a sample. In this case, the antigen is fixed to the slide and dilutions of patient samples are fixed to several slides and incubated in the dark at room temperature to give them time to react with the antigen. The FITC-labeled antibody is an anti-gamma globulin antibody. This means that if there is any patient antibody bound to antigen on the slide, the fluorescent antibody will bind to it. The slides are rinsed to remove any unbound fluorescent antibodies. The slides are now ready to view under the microscope with a UV light source. If the antibody is present, the labeled antibodies will fluoresce a bright apple green.
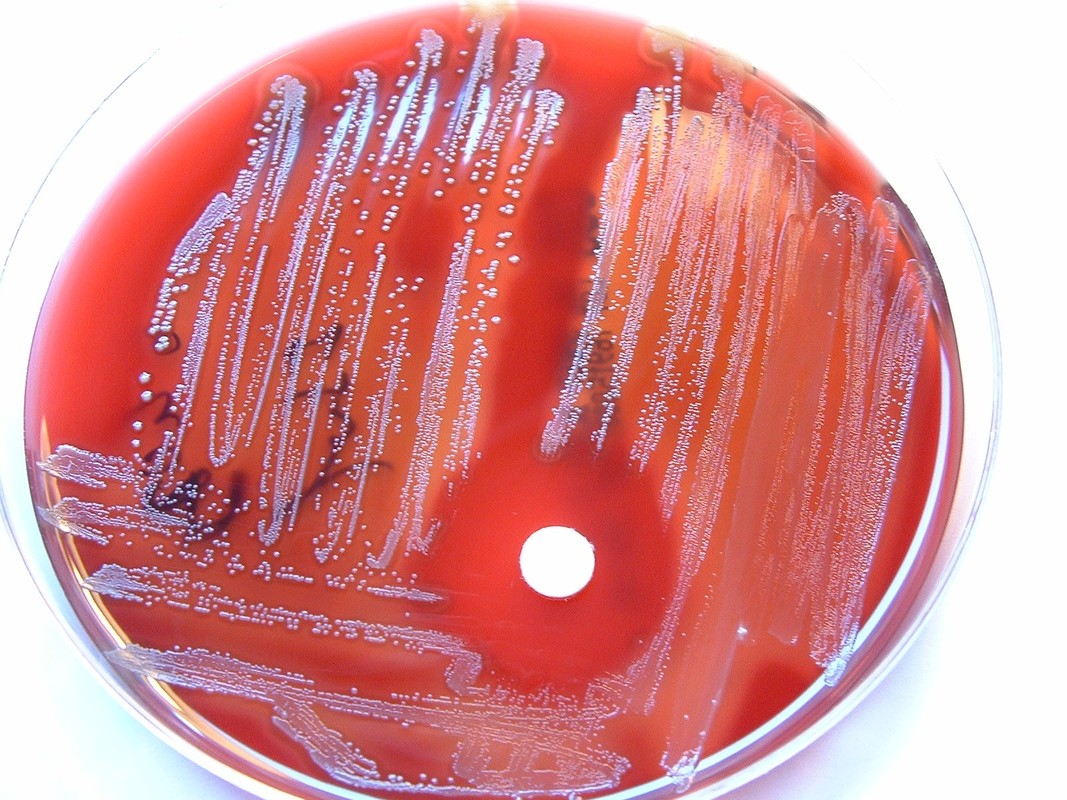
hemolysis patterns on 5% trypticase soy sheep's blood agar:
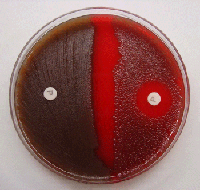
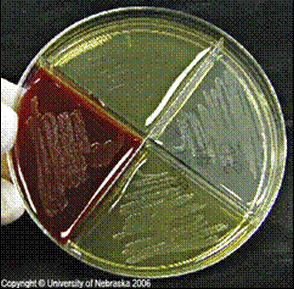
The Sheep Blood Agar plate in the image above shows examples of alpha hemolysis (left), beta hemolysis (middle) and gamma hemolysis (right).
HEMOLYSIS PATTERNS:
Hemolysis defined is a breakdown of red blood cells. Microbiologists are familiar with the hemolysis patterns of microorganisms, and the ability of various bacterial colonies to induce hemolysis when they are grown on blood agar. This aids the microbiologist in the classification of certain microorganisms based on their hemolysis patterns, along with other tests. An enzyme that is able to induce hemolysis is referred to as a hemolysin. There are three types of hemolysis: alpha hemolysis (a-hemolysis), beta hemolysis (B-hemolysis) and gamma hemolysis (y-hemolysis).
1) Alpha hemolysis: Turns the agar below the colony a dark, greenish color. Other synonymous terms are incomplete hemolysis or partial hemolysis, because there is only a partial hemolysis of the red blood cells in the agar surrounding the bacterial colonies. Some bacteria produce hydrogen peroxide, which basically oxidizes the hemoglobin carried by the red blood cells in the blood agar to methemoglobin, producing a green coloring of the agar. Streptococcus pneumoniae and Streptococcus viridans are alpha hemolytic (sometimes this one is gamma-hemolytic). A few other examples include Lactobacillus spp and Gardnerella vaginalis (may be alpha or gamma-hemolytic)
2) Beta hemolysis: Produces a zone of clearing underneath and surrounding the bacterial colonies. Beta hemolysis (β-hemolysis) is also referred to as complete hemolysis. It is a complete lysis of red blood cells in the media around and under the colonies, therefore, the area appears lightened (yellow), clear and transparent. An exotoxin called streptolysin is an enzyme produced by the bacteria, responsible for the complete lysis of red blood cells. There are two types of streptolysin: Streptolysin O (SLO) and streptolysin S (SLS). Streptolysin O is an oxygen-labile cytotoxin, secreted by most Group A streptococcus (GAS). It interacts with cholesterol in the membrane of eukaryotic cells, such as the body's red and white blood cells, macrophages, and platelets, and usually results in β-hemolysis under the surface of blood agar because of its ability to lyse the RBCs in the agar. Streptolysin S is an oxygen-stable cytotoxin also produced by most GAS strains, which results in clearing on the surface of blood agar. SLS affects immune cells, including polymorphonuclear leukocytes (PMNs) and lymphocytes, and prevents the host immune system from clearing infection. Streptococcus pyogenes, or Group A beta-hemolytic Strep (GAS), displays beta hemolysis. A few other examples include S. agalactiae, or Group B beta-hemolytic Strep (GBS), Listeria monocytogenes, Bacillus spp, Clostridium spp, and some strains of E. coli.
3) Gamma hemolysis: Does not produce any hemolysis on the agar. In this case, the organism does not induce hemolysis, and the agar under and around the colony is unchanged. The organism is called non-hemolytic or said to display gamma hemolysis (γ-hemolysis). Enterococcus faecalis (formerly called "Group D Strep") displays gamma hemolysis. A few other examples include some of the members of the Coagulase Negative Staphylcocci, such as S. epidermidis, and S. saprophyticus, as well as most of the Corynebacterium spp, and some of the members of the Enterobacteriaceae.
TIP FOR STUDENTS AND NEW MICROBIOLOGISTS: It is helpful to hold the plate up to the light in order to clearly see the patterns of hemolysis and look at both the surface of the plate AND the underside of the plate in the light. Sometime it may be very difficult to see, particularly if there is a single colony that displays beta hemolysis, or if a beta hemolytic colony is growing either on top of or underneath other types of colonies, such as alpha hemolytic ones. This takes practice and with time, can be more easily differentiated.
HEMOLYSIS PATTERNS:
Hemolysis defined is a breakdown of red blood cells. Microbiologists are familiar with the hemolysis patterns of microorganisms, and the ability of various bacterial colonies to induce hemolysis when they are grown on blood agar. This aids the microbiologist in the classification of certain microorganisms based on their hemolysis patterns, along with other tests. An enzyme that is able to induce hemolysis is referred to as a hemolysin. There are three types of hemolysis: alpha hemolysis (a-hemolysis), beta hemolysis (B-hemolysis) and gamma hemolysis (y-hemolysis).
1) Alpha hemolysis: Turns the agar below the colony a dark, greenish color. Other synonymous terms are incomplete hemolysis or partial hemolysis, because there is only a partial hemolysis of the red blood cells in the agar surrounding the bacterial colonies. Some bacteria produce hydrogen peroxide, which basically oxidizes the hemoglobin carried by the red blood cells in the blood agar to methemoglobin, producing a green coloring of the agar. Streptococcus pneumoniae and Streptococcus viridans are alpha hemolytic (sometimes this one is gamma-hemolytic). A few other examples include Lactobacillus spp and Gardnerella vaginalis (may be alpha or gamma-hemolytic)
2) Beta hemolysis: Produces a zone of clearing underneath and surrounding the bacterial colonies. Beta hemolysis (β-hemolysis) is also referred to as complete hemolysis. It is a complete lysis of red blood cells in the media around and under the colonies, therefore, the area appears lightened (yellow), clear and transparent. An exotoxin called streptolysin is an enzyme produced by the bacteria, responsible for the complete lysis of red blood cells. There are two types of streptolysin: Streptolysin O (SLO) and streptolysin S (SLS). Streptolysin O is an oxygen-labile cytotoxin, secreted by most Group A streptococcus (GAS). It interacts with cholesterol in the membrane of eukaryotic cells, such as the body's red and white blood cells, macrophages, and platelets, and usually results in β-hemolysis under the surface of blood agar because of its ability to lyse the RBCs in the agar. Streptolysin S is an oxygen-stable cytotoxin also produced by most GAS strains, which results in clearing on the surface of blood agar. SLS affects immune cells, including polymorphonuclear leukocytes (PMNs) and lymphocytes, and prevents the host immune system from clearing infection. Streptococcus pyogenes, or Group A beta-hemolytic Strep (GAS), displays beta hemolysis. A few other examples include S. agalactiae, or Group B beta-hemolytic Strep (GBS), Listeria monocytogenes, Bacillus spp, Clostridium spp, and some strains of E. coli.
3) Gamma hemolysis: Does not produce any hemolysis on the agar. In this case, the organism does not induce hemolysis, and the agar under and around the colony is unchanged. The organism is called non-hemolytic or said to display gamma hemolysis (γ-hemolysis). Enterococcus faecalis (formerly called "Group D Strep") displays gamma hemolysis. A few other examples include some of the members of the Coagulase Negative Staphylcocci, such as S. epidermidis, and S. saprophyticus, as well as most of the Corynebacterium spp, and some of the members of the Enterobacteriaceae.
TIP FOR STUDENTS AND NEW MICROBIOLOGISTS: It is helpful to hold the plate up to the light in order to clearly see the patterns of hemolysis and look at both the surface of the plate AND the underside of the plate in the light. Sometime it may be very difficult to see, particularly if there is a single colony that displays beta hemolysis, or if a beta hemolytic colony is growing either on top of or underneath other types of colonies, such as alpha hemolytic ones. This takes practice and with time, can be more easily differentiated.
oxidase test (dry slide/tube/spot):
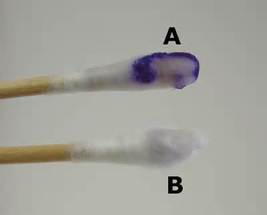
OXIDASE TEST:
Our site uses the filter paper method, but other hospitals and sites may use different kits or techniques, as shown below.
Principle of the Test:
This test is performed to distinguish among groups of bacteria on the basis of cytochrome oxidase activity. During aerobic respiration, oxidase plays a key role in the operation of the electron transport chain (ETC). In fact, an enzyme called cytochrome oxidase catalyzes ("kicks off") the reaction. It catalyzes the oxidation of a reduced cytochrome by molecular oxygen (O2). This results in the formation of water or hydrogen peroxide. Aerobic bacteria, some facultative anaerobes, and microaerophiles display oxidase activity. This test aids in distinguishing among members of the generas Neisseria spp, Haemophilus spp, Moraxella catarrhalis, and Pseudomonas aeruginosa, both of which are oxidase-positive, and most of the Enterobacteriaceae, which are oxidase-negative. These are just a few examples of oxidase + organisms.
To perform the test, depending upon the method your laboratory uses, follow the steps listed under the images below. Each laboratory is different and may use slightly different methods, but the principle is the same.
Our site uses the filter paper method, but other hospitals and sites may use different kits or techniques, as shown below.
Principle of the Test:
This test is performed to distinguish among groups of bacteria on the basis of cytochrome oxidase activity. During aerobic respiration, oxidase plays a key role in the operation of the electron transport chain (ETC). In fact, an enzyme called cytochrome oxidase catalyzes ("kicks off") the reaction. It catalyzes the oxidation of a reduced cytochrome by molecular oxygen (O2). This results in the formation of water or hydrogen peroxide. Aerobic bacteria, some facultative anaerobes, and microaerophiles display oxidase activity. This test aids in distinguishing among members of the generas Neisseria spp, Haemophilus spp, Moraxella catarrhalis, and Pseudomonas aeruginosa, both of which are oxidase-positive, and most of the Enterobacteriaceae, which are oxidase-negative. These are just a few examples of oxidase + organisms.
To perform the test, depending upon the method your laboratory uses, follow the steps listed under the images below. Each laboratory is different and may use slightly different methods, but the principle is the same.
indole test (dry slide/tube/spot):
Tryptophan is an essential amino acid that some bacteria utilize by oxidizing it with the release of enzymes and by converting it into metabolic products mediated by the enzyme tryptophanase. This ability to hydrolyze tryptophan results in the production of pyruvic acid, ammonia and indole. The presence of indole is detectable via a color indicator. Kovac's reagent produces a cherry red color. Other formulations produce a blue color. The color is produced by the reagent, composed of p-dimethylaminobenzaldehyde, butanol, and hydrochloric acid. E. coli is an example of an indole + organism. A few more examples include Proteus vulgaris, Klebsiella oxytoca, Aeromonas hydrophila.
cefinase disk diffusion test (beta-lactamase/nitrocefin):
This test is a disk test used to detect the presence of the enzyme beta lactamase, produced by some microbes such as Staphylococcus aureus, Neisseria gonorrhoeae, and Enterococcus species. It is used to see if the specimen is resistant to penicillin and the cephalosporins. In this test, a drop of water is added to a slide to hold the disk in place. A loopful of test bacteria is smeared onto the surface of the disk. Positive organisms will produce a red-pink color within 5 minutes. If the test is negative, no color change will occur.
Penicillins and the cephalosporins are called B-lactam antibiotics. This is because they have a B-lactam ring in their chemical formula that kill bacteria by stopping cell wall synthesis. The bacteria contain enzymes, such as transpeptidase, that cause peptidoglycan to cross-link each other, making a sturdy, rigid cell wall. The B-lactam antibiotics bind here instead, preventing the cell wall from forming. Some bacteria have developed resistance to this mechanism by producing enzymes called B-lactamases, which hydrolyze the B-lactam ring, destroying the antibiotic and pumping it out. The B-lactamase test is used to detect formation of this enzyme.
Beta-lactamase enzymes serve a crucial role in bacterial resistance to beta-lactam antimicrobials. It is important to detect their presence, because beta-lactamases cut open the beta-lactam ring, which is a part of penicillin antibiotics. By using a chromogenic cephalosporin to serve as a substrate, this test results in a pink-colored disk if the organism is a beta-lactamase producer. Examples of beta-lactamase + organisms include: S. aureus, MRSA, Other Staphylococci, N. gonorrhoeae, H. influenzae, M. catarrhalis, Some enterobacteriaceae, Some strains of P. aeruginosa.
Penicillins and the cephalosporins are called B-lactam antibiotics. This is because they have a B-lactam ring in their chemical formula that kill bacteria by stopping cell wall synthesis. The bacteria contain enzymes, such as transpeptidase, that cause peptidoglycan to cross-link each other, making a sturdy, rigid cell wall. The B-lactam antibiotics bind here instead, preventing the cell wall from forming. Some bacteria have developed resistance to this mechanism by producing enzymes called B-lactamases, which hydrolyze the B-lactam ring, destroying the antibiotic and pumping it out. The B-lactamase test is used to detect formation of this enzyme.
Beta-lactamase enzymes serve a crucial role in bacterial resistance to beta-lactam antimicrobials. It is important to detect their presence, because beta-lactamases cut open the beta-lactam ring, which is a part of penicillin antibiotics. By using a chromogenic cephalosporin to serve as a substrate, this test results in a pink-colored disk if the organism is a beta-lactamase producer. Examples of beta-lactamase + organisms include: S. aureus, MRSA, Other Staphylococci, N. gonorrhoeae, H. influenzae, M. catarrhalis, Some enterobacteriaceae, Some strains of P. aeruginosa.
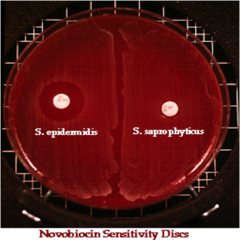
novobiocin disk diffusion test:
Staphylococcus epidermidis is susceptible to Novobiocin.
Staphylococcus saprophyticus is resistant to Novobiocin.
This test is used to differentiate between these 2 coagulase negative staphylococci species. S. saprophyticus is linked to urinary tract infections in young, sexually active females between the ages of 14-29 years of age. S. epidermidis is linked to UTI catheter infections, IV catheter infections, and is the #1 cause of bacterial infection in prosthetic heart valves due to its ability to produce biofilm (acute infectious endocarditis).
bacitracin susceptibility test:
The bacitracin disk test is used to differentiate B-hemolytic group A streptococci from other B-hemolytic streptococci. Place a bacitracin-impregnated disk on an agar plate that has been inoculated to produce a bacterial lawn, and the disk will diffuse into the agar. If its concentration is sufficient, it will inhibit the growth of susceptible bacteria, appearing as a zone of clearing on the agar plate. If the zone of clearing is >10 mm, it is considered as being susceptible to bacitracin.
optochin disk diffusion test (taxo p):
The optochin disk test (TAXO P) is a growth inhibition test in which a 6-mm filter-paper disc impregnanted with 5 mg of ethylhydrocupreine hydrochloride (optochin) and are called P-discs are placed on the surface of a blood agar plate streaked with a lawn of bacterial growth of the test organism. S. pneumoniae is sensitive to this agent, and cells are lysed by the antibiotic, resulting in the formation of a zone of inhibition >=14 mm surrounding the P-disk. Nonpneumoncoccal alpha-hemolytic streptococci are resistant to the optochin disc and fail to show a zone of inhibition or produce a zone of <14 mm.
pyr disk test:
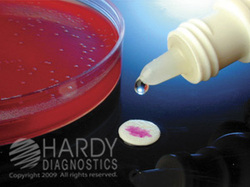
PYR DISK TEST:
In the PYR disk test, a small drop of water is placed on a slide to hold a PYR disk in place. A loopful of test bacteria is added to the disk and allowed to sit at room temperature for 2 minutes. A drop of PYR reagent is then added to the disk after 2 minutes. PYR positive organisms will turn a pink-fuschia-red color within seconds of addition of PYR reagent. PYR negative organisms show no color change. In this test, the enzyme L-pyrroglutamyl-aminopeptidase hydrolyzes the substrate L-pyrrolidonyl-B-naphthylamide (PYR) resulting in the production of B-naphthylamine, which combines with a cinnamaldehyde reagent, producing a bright red color. This test is useful in identifying Gram-positive cocci such as S. pyogenes and Enterococcus spp and S. lugdunensis.
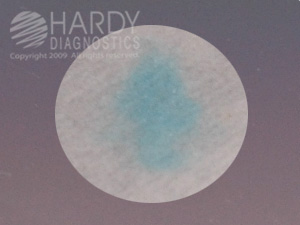
moraxella catarrhalis (mcat) disk test (butyrate):
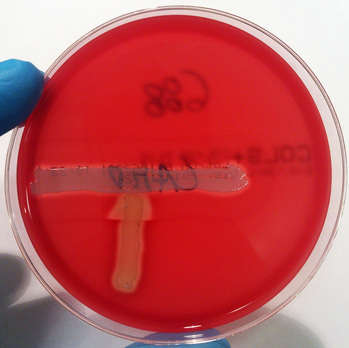
The Moraxella catarrhalis disk test can be used to aid in the identification of this organism. A drop of water is placed on a slide to hold the MCAT disk in place. A loopful of organism is then smeared onto the surface of the disk. Within 5 minutes, the disk will change to a blue color if the organism is M. catarrhalis.
hippurate hydrolysis:
The hippurate hydrolysis test is based on the ability of the enzyme hippuricase to hydrolyze the substrate hippurate to produce the amino acid glycine. Ninhydrin reagent detects oxidized glycine resulting in a deep purple color. It is used to identify the organisms S. agalactiae, Campylobacter jejuni, Gardnerella vaginalis, and Listeria monocytogenes, all of which are positive.
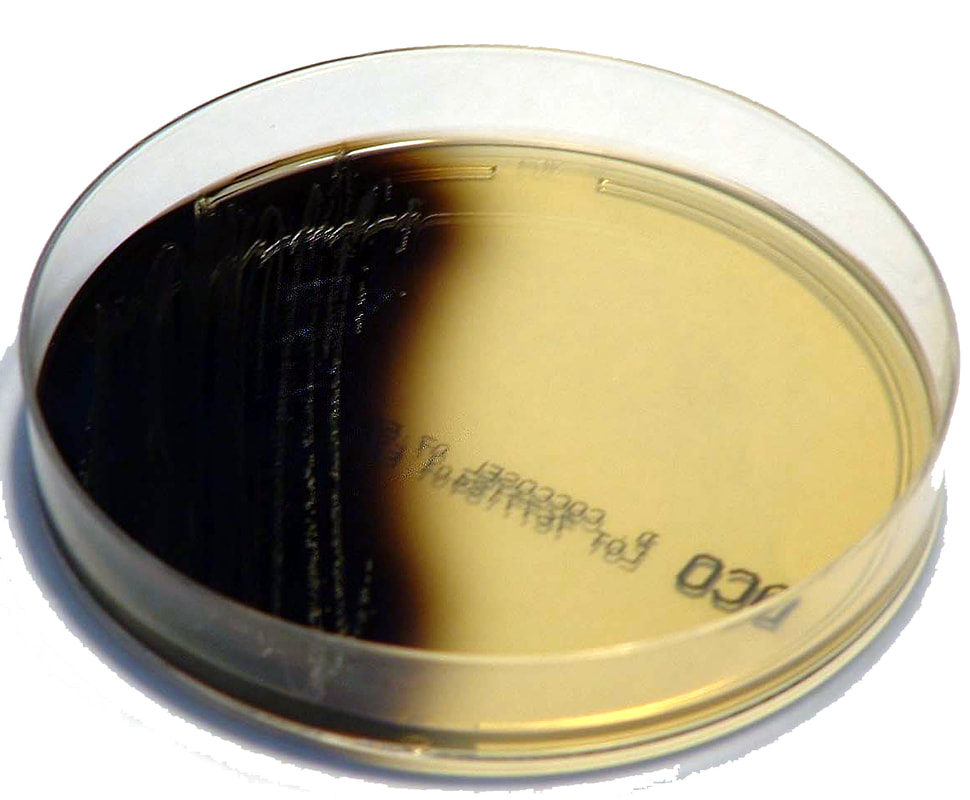
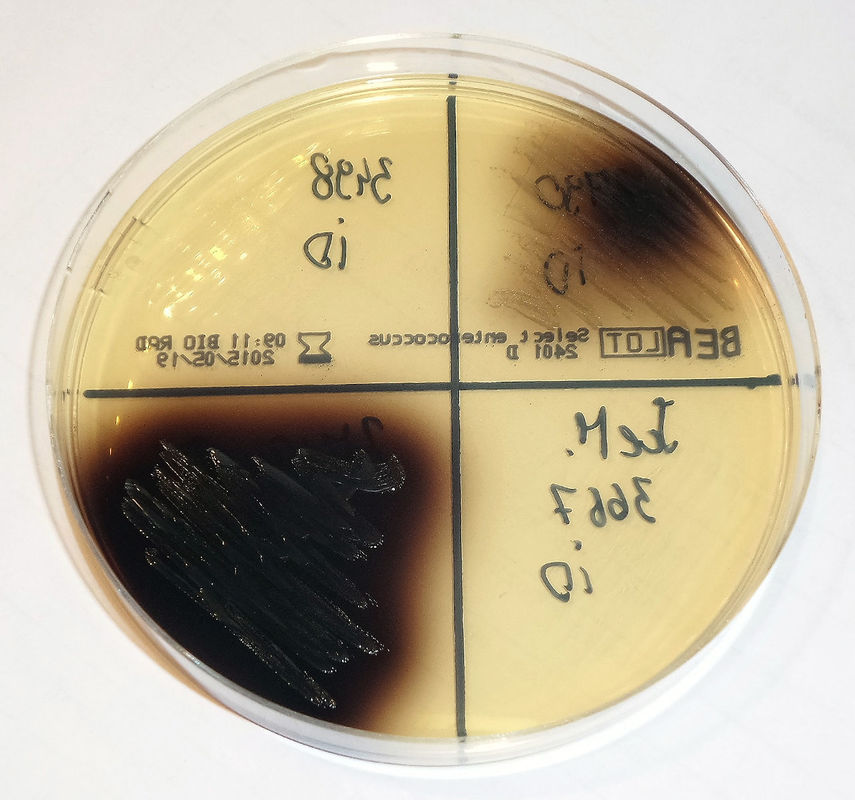
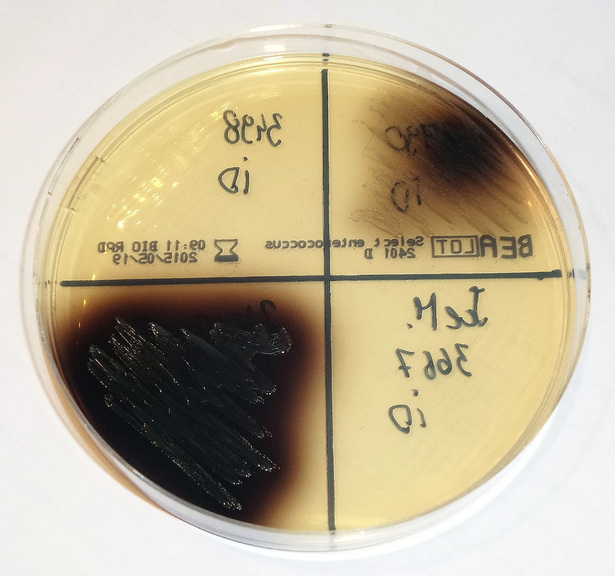
Bile esculin test:
The bile esculin test is a test used to aid in the identification of enterococci and S. bovis group, which are positive for bile esculin hydrolysis. Bile esculin agar is selective and differential and comes in an agar plate or slant. It consists of a peptone base with esculin, bile, sodium azide, and ferric citrate. Esculin provides the glucose sugar, peptone provides proteins, bile blocks growth of Gram-positive organisms other than Group D streptococci and enterococci. Sodium azide blocks the growth of Gram-negative organisms. Ferric citrate is the color indicator. Under acidic conditions, when the bacteria ferment the glucose and esculin, it becomes hydrolyzed in the presence of bile. As the esculin molecules are split, they react with the ferric citrate to form a dark brown-black phenolic iron complex. It will darken in 18-24 hours and if a slant is more than half darkened at any time within that 48 hours, it can be recorded as positive.
triple sugar iron (tsi) agar slants:
This biochemical test is utilized to observe whether or not a Gram-negative bacilli ferment glucose and lactose or sucrose and form H2S or or gas. The media contains 10 parts lactose, 10 parts sucrose, and 1 part glucose and peptone. pH indicators included are phenol red and ferrous sulfate, which detect acidification of the media when sugars are fermented and detect the production of H2S. When glucose is fermented, the whole medium becomes acidic and turns yellow within 8-12 hours. After 18-24 hours, the butt of the tube remains acidic due to the presence of organic acid byproducts from glucose fermentation under anaerobic conditions, however, the slant reverts back to alkaline (red) because of oxidation of the fermentation byproducts under aerobic conditions on the slant. CO2 and H2O and the oxidation of peptones to alkaline amines are responsible for this change. When the one or both of the other sugars are fermented in addition to glucose, the huge amount of fermentation products formed on the slant with neutralize the alkaline amines and cause the slant to become yellow as well (acidic). The reaction MUST be read in 18-24 hours. NOTE: REACTIONS IN TSI SHOULD NOT BE READ >24 HOURS OF INCUBATION DUE TO AEROBIC OXIDATION OF THE FERMENTATION PRODUCTS FROM LACTOSE AND/OR SUCROSE, WHICH WILL REVERT BACK TO THE ALKALINE STATE.
The formation of H2 (hydrogen gas) and CO2 is observable by the formation of bubbles, cracks or movement of the agar by separation of the agar from the bottom and/or sides of the glass tube. Production of H2S demands an acidic atmosphere and is observed by a blackening in the butt of the medium of the tube.
Inoculation of this media is performed at room temperature with a sterilized, straight inoculation needle touched to the top of one well-isolated colony and stabbed through the center of the medium to near the bottom of the tube and by then streaking the slant surface. The cap is replaced loosely and incubated in ambient air for 18-24 hours.
Results:
Alkaline slant/no color change in the butt: glucose, lactose and/sucrose non-fermenter (K/K or K/NC)
Alkaline slant/acid butt: glucose fermentation only (K/A)
Acid slant/acid butt: glucose, sucrose, and/or lactose fermenter (A/A)
Black Precipitate: black precipitate in the butt of the tube indications production of ferrous sulfide and H2S gas
Bubbles or Cracks: bubbles or cracks in the agar indicate production of CO2 or H2 gas
The formation of H2 (hydrogen gas) and CO2 is observable by the formation of bubbles, cracks or movement of the agar by separation of the agar from the bottom and/or sides of the glass tube. Production of H2S demands an acidic atmosphere and is observed by a blackening in the butt of the medium of the tube.
Inoculation of this media is performed at room temperature with a sterilized, straight inoculation needle touched to the top of one well-isolated colony and stabbed through the center of the medium to near the bottom of the tube and by then streaking the slant surface. The cap is replaced loosely and incubated in ambient air for 18-24 hours.
Results:
Alkaline slant/no color change in the butt: glucose, lactose and/sucrose non-fermenter (K/K or K/NC)
Alkaline slant/acid butt: glucose fermentation only (K/A)
Acid slant/acid butt: glucose, sucrose, and/or lactose fermenter (A/A)
Black Precipitate: black precipitate in the butt of the tube indications production of ferrous sulfide and H2S gas
Bubbles or Cracks: bubbles or cracks in the agar indicate production of CO2 or H2 gas
urease slant:
In the urease test, urease hydrolyzes the substrate urea into ammonia, water and Co2. The presence of urease is detected by the production of ammonia in the medium, which alkalizes it, resulting in the production of a bright fuschia color, detected by the pH color indicator in the medium. The urease test aids in the identification of Enterobacteriaceae, such as Proteus spp, Corynebacterium urealyticum, H. pylori.
Lysine iron agar (lia):
Lysine Iron Agar test is used to observe whether or not a Gram-negative bacilli is able to decarboxylate or deaminate lysine and form H2S. LIA agar contains lysine, peptones, glucose, ferric ammonium citrate and sodium thiosulfate. Upon glucose fermentation, the butt of the medium acidifies and turns yellow. If the organism is a lysine decarboxylater, cadaverine is formed, which neutralizes the organic acids that were formed by glucose fermentation, causing the butt to revert to a purple color (alkaline state). If decarboxylase is not produced, then the butt remains yellow (acidic). If oxidation deamination of lysine occurs, then a flavin mononucleotide compound is formed, resulting in a burgandy color on the slant, whereas if deamination does not occur, then the LIA slant stays purple.
tube motility test:
Motility indole agar or SIM agar can be used to perform this test.
Motility is indicated by haziness/turbidity in the agar and movement away from the stab line, as seen in the image below. Umbrella motility is displayed by Listeria monocytogenes. Non-motile organisms do not show movement away from the stab line, and the stab line will remain clearly visible, with clear agar.
Motility is indicated by haziness/turbidity in the agar and movement away from the stab line, as seen in the image below. Umbrella motility is displayed by Listeria monocytogenes. Non-motile organisms do not show movement away from the stab line, and the stab line will remain clearly visible, with clear agar.
Bile esculin test:
The bile-esculin test is used a lot for the differentiation of enterococci and Group D streptococci from non-Group D viridans streptococci. Enterococci and Group D streptococci are bile-tolerant and can hydrolyze esculin in the presence of bile (4% bile salts or 40% bile). The sensitivity and specificity are >90%. A positive bile esculin test will cause the agar to blacken because esculetin reacts with ferric ions supplied in the agar to form a black, diffusible complex. E. faecalis, E. faecium, S. bovis, Listeria monocytogenes are some of the BEA positive organisms. This can be performed on BEA plates or slants.
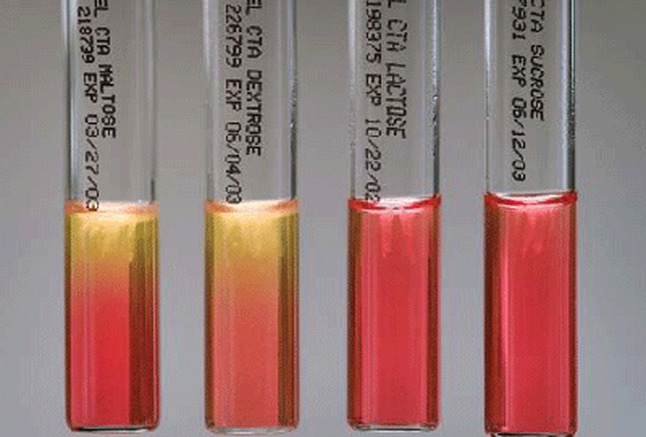
The Sugars Test:
4 Slants: Glucose (Dextrose), Sucrose, Lactose and Maltose
The leucine aminopeptidase disk test (lap):
This is a disk test by Remel used to detect enzymatic production of leucine aminopeptidase by streptococci and other related species. Filter paper disks are impregnated with the substrate leucine-B-naphthylamide hydrochloride. If the organism produces this enzyme, then the substrate is hydrolyzed to release leucine and B-naphthylamide. After addition of 1 drop of color developer, the B-naphthylamide then combines with the color developer (cinnamaldehyde) to form a pink-red color on the disk.
To perform the test, catalase negative, Gram-positive cocci are smeared onto a disk slightly moistened with sterile water and allowed to incubate at room temperature for 5 minutes. 1 drop of PYR color reagent is added to the disk and read immediately.
LAP positive organisms include the following:
Aerococcus urinae
Enterococcus spp
Lactococcus spp
Pediococcus spp
Streptococcus spp
LAP negative organisms include the following:
Other Aerococcus spp
Leuconostoc spp
To perform the test, catalase negative, Gram-positive cocci are smeared onto a disk slightly moistened with sterile water and allowed to incubate at room temperature for 5 minutes. 1 drop of PYR color reagent is added to the disk and read immediately.
LAP positive organisms include the following:
Aerococcus urinae
Enterococcus spp
Lactococcus spp
Pediococcus spp
Streptococcus spp
LAP negative organisms include the following:
Other Aerococcus spp
Leuconostoc spp
onpg test:
The ONPG test allows organisms to be classified as a lactose fermenter by testing for beta-galactosidase.
India Ink test:
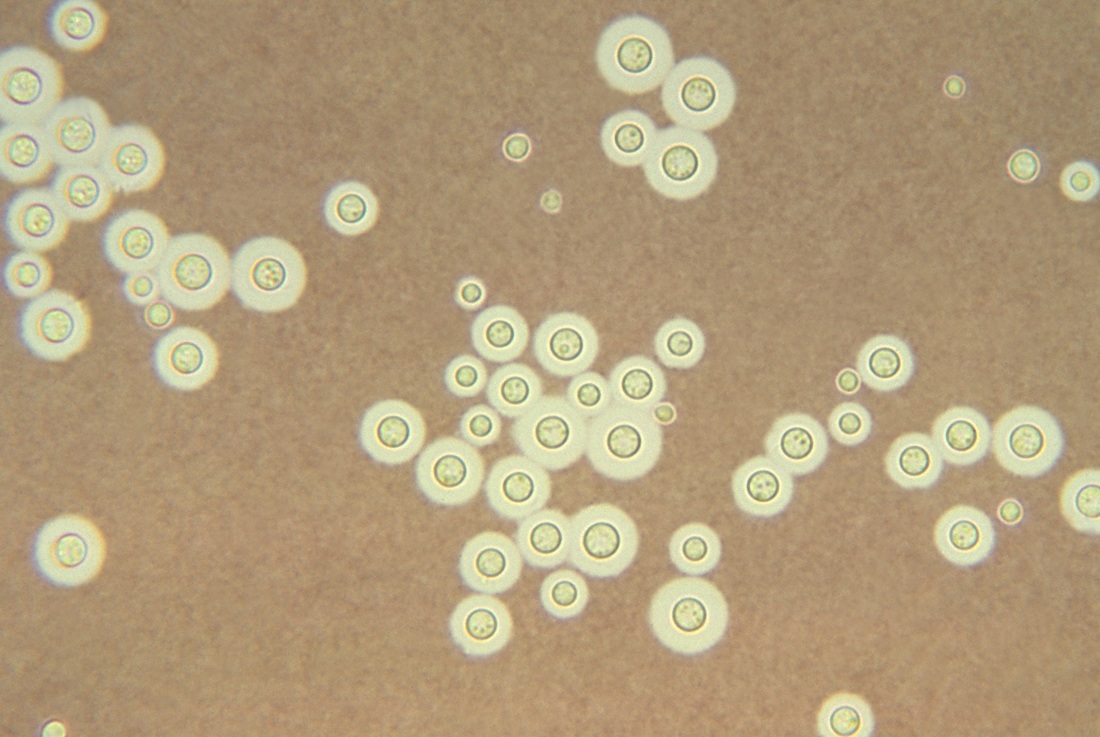
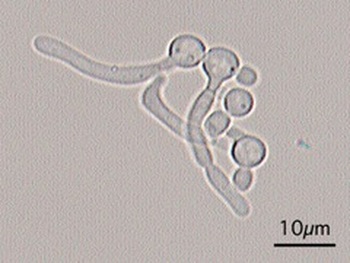
This test is often used to detect Cryptococcus neoformans in CSF and will show the capsule around the yeast. After mixing the India Ink tube well, add one drop to a clean slide. Add a drop of CSF (centrifuged, supernatant poured off, and test the button) and carefully mix with the edge of a flamed coverslip, adding the coverslip carefully. View under the 10x and 40x objective. A positive test will show a complete clear zone around this yeast. Note that other yeasts and white blood cells will also stand out against the dark background, but C. neoformans is the only one with a thick, clear capsule surrounding it. Also note that Brownian motion will cause the black India Ink particles to appear as if they are moving.
Germ Tube test:
This test differentiates Candida albicans yeast from other Candida spp because C. albicans is the only one that is positive for formation of germ tubes after 2-2.5 hours of incubation at 37 degrees Celsius. After incubation, add one drop of solution to the slide with a coverslip and view under low condenser light under 20x and 40x objective.
IMViC reaction:
The IMViC reaction differentiates E. coli from Enterobacter spp. https://en.wikipedia.org/wiki/IMViC
I=Indole Test
M=Methyl Red Test
V=Voges-Proskauer Test
i=in
C=Citrate Agar
I=Indole Test
M=Methyl Red Test
V=Voges-Proskauer Test
i=in
C=Citrate Agar
CAMP test:
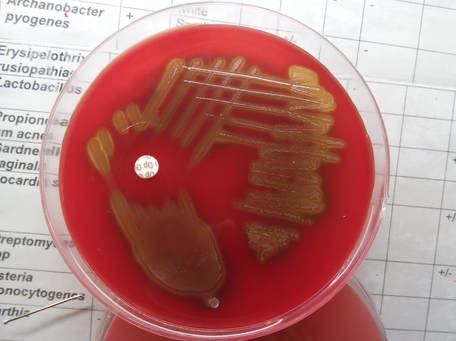
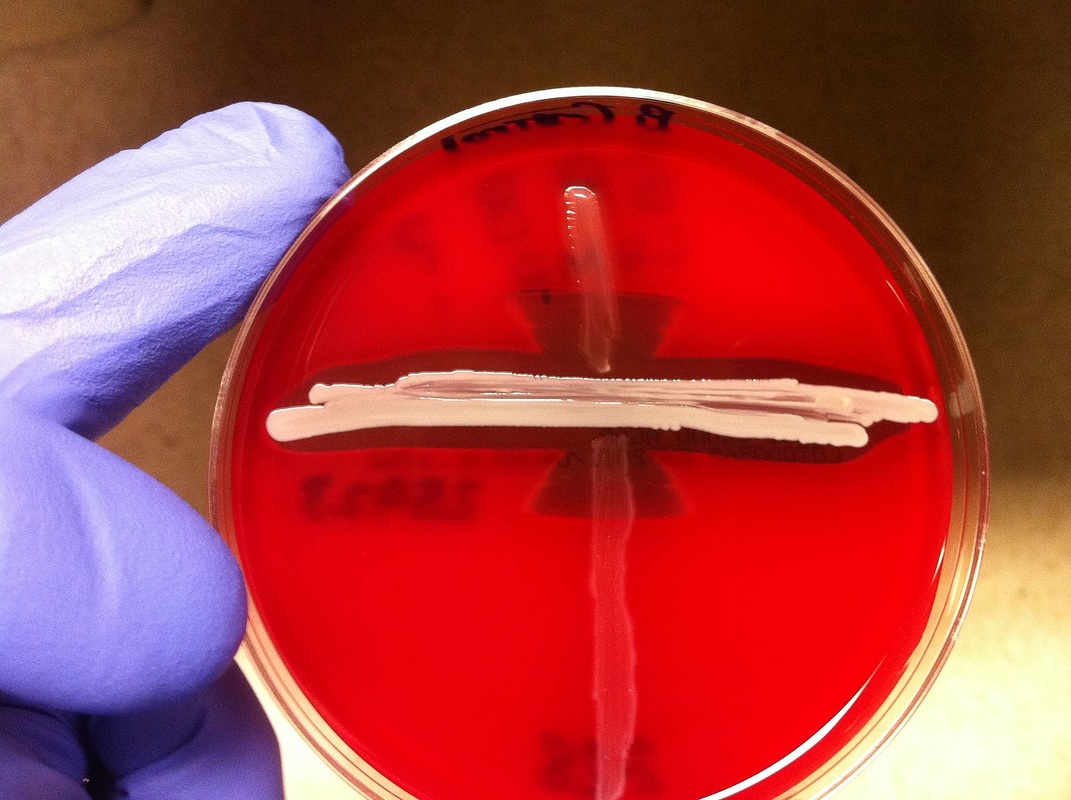
This test is performed for the detection of Group B streptococci (S. agalactiae) when the Pdx latex test is negative, and there is beta hemolysis resembling GBS. To perform this test, make a streak straight across the plate with S. aureus. At a right angle to, but not touching the S. aureus streak, make streaks of the test organism. If the organism is S. agalactiae, it will produce zones of clearing shaped like "arrows" or "wedges"Th pointing toward the S. aureus streak, as seen in the images below. This test is used to identify Group B streptococci based on their production of CAMP factor. This factor enlarges the hemolysis zone that is produced by S. aureus's release of the enzyme B-hemolysin.
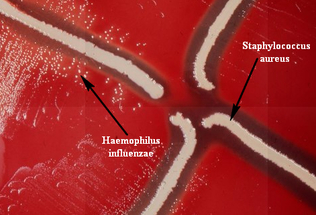
satellite test:
The satellite test is used to isolate H. influenzae, which grows or "satellites" in the presence of S. aureus on sheep's blood agar in the zones of hemolysis where factor V has been released (H. influenzae needs this factor to grow). See the images below.
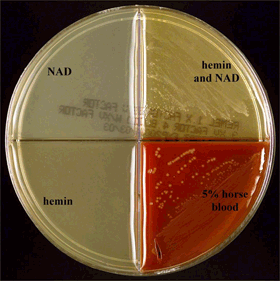
Haemophilus quad plate:
H. influenza only grows in the quadrants containing hemin and NAD and the one containing the 5% horse blood, but it will not hemolyze the horse blood. H. haemolyticus also grows in these two quadrants. In order to differentiate between the two species, you need to check the hemolysis type on sheep or horse blood agar in that particular quadrant. An organism growing in JUST the NAD OR JUST the hemin quadrant means that it is another species of Haemophilus, not influenzae or haemolyticus. Also, if a species is growing in EVERY quadrant, it is not likely H. haemophilus species. Every once in awhile, a H. influenzae spp organism will show slight growth in the NAD quadrant.
- H. influenzae: growth in hemin AND NAD quad; growth in horse blood quad; no hemolysis
- H. haemolyticus: growth in hemin AND NAD quad; growth in horse blood quad; hemolysis of blood agar
- H. parahaemolyticus: growth in NAD quad, NAD + hemin quad, and horse blood quad; hemolysis of blood agar