PCR: Polymerase chain reaction
Biofire film array diagnostics:
The BioFire Film Array PCR is the newest PCR technology in molecular infectious disease diagnostics. It is efficient, quick and cost-effective. Plus, it can test multiplex panels of diseases simultaneously, proving it is comprehensive, and it is also quite accurate. The testing saves space, requires little sample, is simple and easy to use, and is able to interface with laboratory LIS systems. It takes as little as 2 minutes of prep time (hands-on) and is user friendly. Physicians and healthcare workers love this technology because it results in faster turnaround time, quicker and more precise treatments, and shorter length of hospital stays for the patient. Because the panels are able to test numerous assays at once, the workflow is quicker and more efficient, less tests need to be performed, the prescribed antibiotics are more accurate, and infection control is improved, resulting in better patient care.
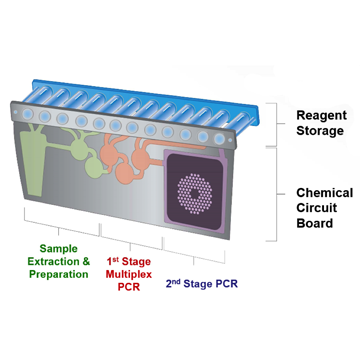
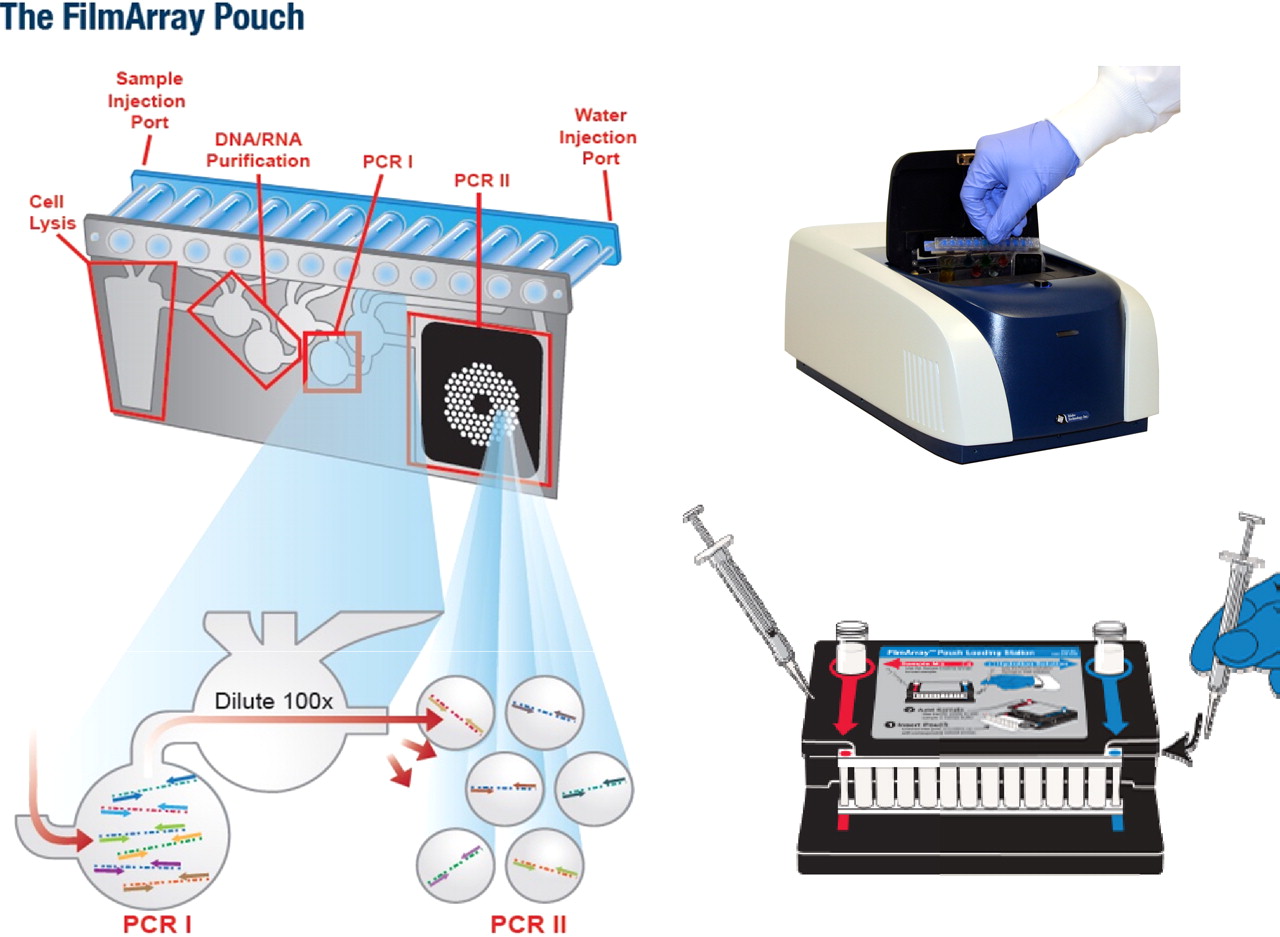
The film array pouches are self-contained, and include the reagents needed to prepare the sample, run PCR, run reverse-transcription PCR (amplification) and detect and identify the microbe. It is able to extract and purify the nucleic acids and perform the PCR testing in one run. The software performs the calculations, graphs and charts necessary to display the result for each target. There are 12 wells containing freeze-dried reagent attached to a chemical circuit board. This is a type of real-time PCR.
The first-stage is a multiplex PCR and the pouch contains 70-90 primers for this stage. This includes 20-25 cycles and then dilutes the sample by 100-fold. The loud buzzing sound during this stage is due to the beads being moved around as cells are lysed to release genetic material. The 2nd-stage PCR is a single stage PCR that purifies the sample. The reagents move through the pouch via air-filled blisters that inflate and pistons to open and close the channels, along with plungers that deliver the reagents where they need to go. The organism(s) that are detected, if any, print out on a report all in about an hour. CLIA states this is a moderate complexity test.
Here is a link to a great video webinar (free) that explains this technology: https://vimeo.com/104643565
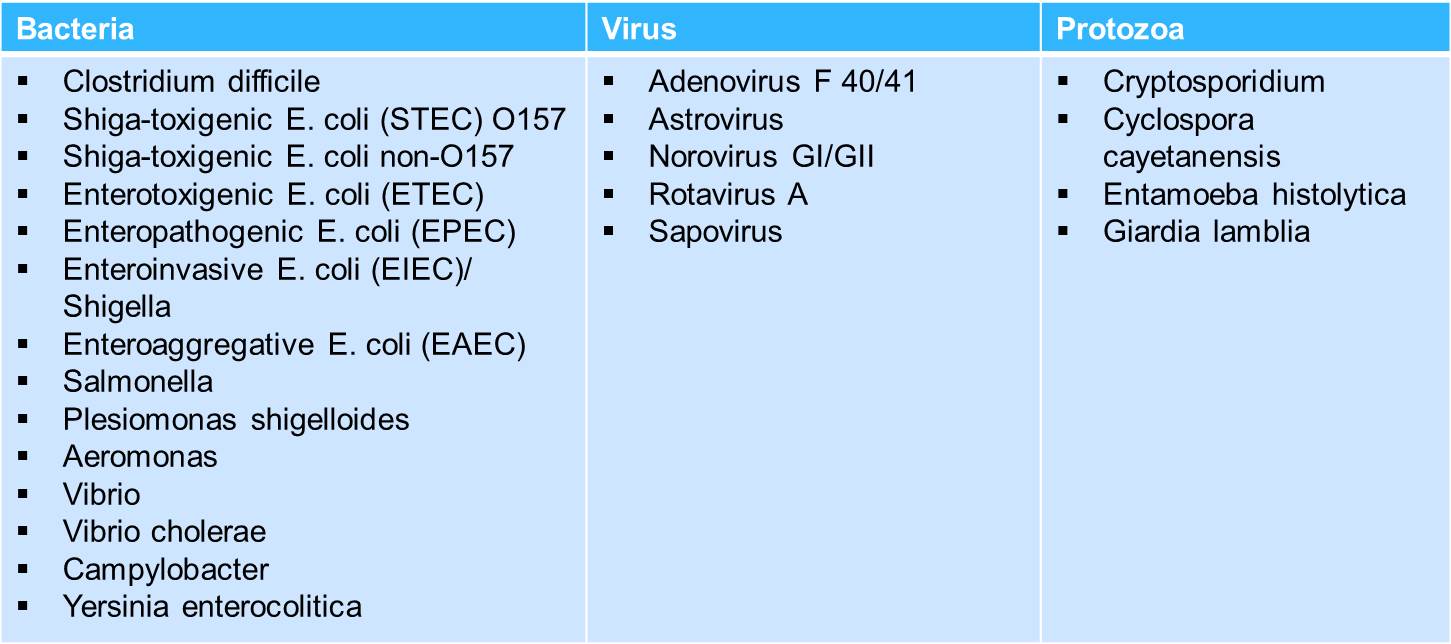
Biofire Filmarray consists of several panels to choose from for testing, including the respiratory panel, GI panel, meningitis panel, and blood culture panel.
Prior to beginning this procedure, don appropriate PPE and always change gloves between samples. Wipe the sample port stage and the work area thoroughly with bleach or a bleach wipe. Rinse with DI water, an alcohol wipe or an H2O2 wipe as a rinse to rinse the bleach since it is corrosive. This disinfects the area and prevents the possibility of a false positive or carryover due to leftover DNA/RNA on any spills or drops that may have occurred. Avoid touching the reaction areas with your hands or gloves. Treat the pouch and its contents as infectious material. Discard any used pouches and materials in a biohazard bin.
The film array pouches are self-contained, and include the reagents needed to prepare the sample, run PCR, run reverse-transcription PCR (amplification) and detect and identify the microbe. It is able to extract and purify the nucleic acids and perform the PCR testing in one run. The software performs the calculations, graphs and charts necessary to display the result for each target. There are 12 wells containing freeze-dried reagent attached to a chemical circuit board. This is a type of real-time PCR.
- In the first step, once the sterile water (blue cap) and sample/buffer solution (red cap) are injected into the biochemical reaction tubes, the cells in the sample are lysed (burst open) to release the genetic material. The water hydrates the freeze-dried reagent.
- The next step involves the DNA/RNA purification once the pouch is put onto the machine.
- The third step is multiplex PCR I, in which the sample is diluted even further (x100) and the genetic material is broken down.
- The fourth step is a single-stage PCR II, in which the genetic material is further broken down, purified and the target sequence is identified.
The first-stage is a multiplex PCR and the pouch contains 70-90 primers for this stage. This includes 20-25 cycles and then dilutes the sample by 100-fold. The loud buzzing sound during this stage is due to the beads being moved around as cells are lysed to release genetic material. The 2nd-stage PCR is a single stage PCR that purifies the sample. The reagents move through the pouch via air-filled blisters that inflate and pistons to open and close the channels, along with plungers that deliver the reagents where they need to go. The organism(s) that are detected, if any, print out on a report all in about an hour. CLIA states this is a moderate complexity test.
Here is a link to a great video webinar (free) that explains this technology: https://vimeo.com/104643565
Biofire Filmarray consists of several panels to choose from for testing, including the respiratory panel, GI panel, meningitis panel, and blood culture panel.
Prior to beginning this procedure, don appropriate PPE and always change gloves between samples. Wipe the sample port stage and the work area thoroughly with bleach or a bleach wipe. Rinse with DI water, an alcohol wipe or an H2O2 wipe as a rinse to rinse the bleach since it is corrosive. This disinfects the area and prevents the possibility of a false positive or carryover due to leftover DNA/RNA on any spills or drops that may have occurred. Avoid touching the reaction areas with your hands or gloves. Treat the pouch and its contents as infectious material. Discard any used pouches and materials in a biohazard bin.
genexpert:
The GenEXPERT is a moderately complex real-time PCR analyzer. It is fast, efficient, flexible, the cartridges are self-contained with almost everything needed and required for detection and to run PCR. You can scan it, load it quickly and walk away. Most tests take about 30 min. to 1.5 hours to run. This turnaround time enables physicians to quickly see if a patient has an illness, be treated, and if they need to be admitted to the hospital or put on precautions. It is quick, highly automated, and highly accurate.
The cartridges used consist of 5 parts:
Benefits of this type of technology include a decrease in MRSA infections, a decrease in ICU/CCU admissions and/or length of stay, saving a lot of lives, saving the hospital a lot of money.
Timing of the tests are estimated as follows, once on the instrument:
The cartridges used consist of 5 parts:
- the foot,
- the valve,
- the body,
- the reaction tube, and
- the lid
- Processing chambers:
- Holds the sample
- Holds the reagent (reaction beads) and rinse
- Holds the processed sample
- Holds the waste when testing is complete
- Contains an air chamber to equalize pressure
- A plunger is inserted, which draws sample and fluids
- Rotating valve body:
- This part rotates so that the fluid can move and enter different chambers and to a reaction tube
- What happens here?
- Specimen is isolated on a filter on the bottom of the cartridge
- PCR inhibitors are taken away
- Samples are lysed (ultrasonically), releasing the genetic material as the sample is broken apart
- Sample is mixed with the PCR reagents
- Processed sample is now moved to the integration reaction tube and drawn into a chamber, where it is illuminated by 7-10 colors of LED lights (chemiluminescence)
- Reaction Tube:
- What happens here?
- Rapid thermal cycling in the reaction tube (rapid cooling and heating)
- Excitation of electrons optically (illumination by 7-10 LED colors of light)
- Detection of the target sequence
- What happens here?
Benefits of this type of technology include a decrease in MRSA infections, a decrease in ICU/CCU admissions and/or length of stay, saving a lot of lives, saving the hospital a lot of money.
Timing of the tests are estimated as follows, once on the instrument:
- MRSA for surveillance testing: 45 minutes
- Detects >195 strains
- Primers detect mecA and mecC strains
- S. aureus and MRSA testing for pre-surgical patients, wound patients, or those with positive blood cultures: 1 hour
- It is helpful to know if a patient is colonized with S. aureus since they are 9x higher at risk for developing an infection at a surgical site or if they are on dialysis
- >90% of nosocomial S. aureus-related infections are caused by the patient's own flora they're colonized with
- C. difficile testing: 30-45 minutes
- Norovirus: about in hour
- Group A Strep: 30 minutes
- Influenza A/B: 20-30 minutes
- Enterovirus: 2.5 hours
- Chlamydia and Gonorrhea: 1.5 hours
- Group B Strep: 35 minutes
- Factor Testing: 30 minutes